QLF-D Protocol for Orthodontic Plaque Assessment: A Comprehensive Clinical Framework for Research and Drug Development
This article presents a detailed clinical protocol for implementing Quantitative Light-induced Fluorescence-Digital (QLF-D) technology in orthodontic research and drug development.
QLF-D Protocol for Orthodontic Plaque Assessment: A Comprehensive Clinical Framework for Research and Drug Development
Abstract
This article presents a detailed clinical protocol for implementing Quantitative Light-induced Fluorescence-Digital (QLF-D) technology in orthodontic research and drug development. Targeting researchers, scientists, and pharmaceutical professionals, it explores the scientific principles of QLF-D for quantifying early plaque formation and cariogenic activity around brackets and wires. The protocol encompasses foundational theory, standardized methodological workflows, common troubleshooting scenarios with optimization strategies, and rigorous validation against established metrics. This framework aims to establish QLF-D as a precise, reproducible, and sensitive tool for evaluating anti-plaque agents, biomaterials, and preventive interventions in orthodontic clinical trials, advancing objective endpoints in oral healthcare product development.
The Science Behind QLF-D: Principles and Rationale for Orthodontic Plaque Quantification
Within the framework of developing a standardized Quantitative Light-induced Fluorescence - Digital (QLF-D) clinical protocol for plaque assessment during orthodontic research, understanding the underlying fluorescence signals is paramount. Fixed orthodontic appliances create significant biofilm retention sites, altering plaque ecology. QLF-D technology exploits the natural autofluorescence of dental biofilms and the specific red fluorescence emitted by certain bacterial metabolites to provide a non-invasive, quantitative assessment of plaque maturity, metabolic activity, and caries risk. This application note details the core technology, protocols, and reagent tools for researchers investigating biofilm dynamics in orthodontic patients.
Core Photobiological Principles
Autofluorescence (Green Signal)
Healthy tooth enamel and early, thin dental biofilms exhibit strong natural autofluorescence under violet-blue light (typically 405 nm), emitting in the green spectrum (~520 nm). This signal originates primarily from bacterial and host-derived fluorophores like flavin adenine dinucleotide (FAD) and collagen cross-links. As plaque matures, this signal diminishes due to light scattering and absorption.
Red Fluorescence (Red Signal)
Specific bacterial metabolites within mature, cariogenic biofilms produce a characteristic red fluorescence emission (>630 nm) when excited at 405 nm. The primary fluorophore is a family of compounds called porphyrins, notably protoporphyrin IX and coproporphyrin, which are intermediates in the heme synthesis pathway. These accumulate in anaerobic, metabolically active environments dominated by bacteria like Porphyromonas gingivalis, Prevotella spp., and Actinomyces spp. The red/green fluorescence ratio (R/G value) is a key quantitative metric for plaque pathogenicity.
Table 1: Key Fluorescence Signatures in Dental Biofilms
| Fluorescence Type | Excitation (nm) | Emission Peak (nm) | Primary Fluorophore Source | Associated Biofilm State | Typical QLF-D Ratio Metric |
|---|---|---|---|---|---|
| Green Autofluorescence | 405 | ~520 nm | FAD, Collagen Cross-links | Early/Thin Plaque, Sound Enamel | ΔR (Loss of Green Reflectance) |
| Red Fluorescence | 405 | >630 nm (peak ~630-690) | Porphyrins (Proto- & Coproporphyrin) | Mature, Cariogenic, Anaerobic Plaque | ΔR (Increase in Red Fluorescence) |
| Combined Diagnostic Signal | 405 | Green & Red Channels | Composite of above | Overall Plaque Activity | Red/Green Ratio (R/G) |
Experimental Protocols for In Vitro Validation
Protocol 3.1: Cultivation of Standardized Biofilms for Fluorescence Analysis
Objective: To generate in vitro biofilms with defined red fluorescence characteristics for QLF-D device calibration and experimental intervention testing. Materials: See Scientist's Toolkit (Section 6). Procedure:
- Inoculum Preparation: Grow reference strains (e.g., Streptococcus mutans UA159, Porphyromonas gingivalis ATCC 33277) in appropriate broth anaerobically (80% N₂, 10% H₂, 10% CO₂) at 37°C to mid-log phase.
- Hydroxyapatite Disc Coating: Sterilize saliva-coated hydroxyapatite (sHA) discs. Coat with sterile, clarified human saliva for 1 hour at 37°C.
- Biofilm Growth: Place sHA discs in a 24-well plate. Add 1.5 mL of defined medium (e.g., McBain with 1% sucrose) inoculated with a mixed bacterial suspension (10⁶ CFU/mL total). Refresh medium every 24 hours.
- Incubation: Incubate anaerobically at 37°C for periods ranging from 24h (early biofilm, low red fluorescence) to 120h (mature, high red fluorescence).
- Harvesting: At designated timepoints, gently rinse discs twice in 0.9% saline to remove loosely adherent cells.
Protocol 3.2: Quantitative Fluorescence Imaging and Analysis
Objective: To acquire and quantify green and red fluorescence signals from experimental biofilms. Procedure:
- QLF-D Image Acquisition: Mount biofilm samples in a standardized, dark chamber. Acquire images using a QLF-D device (e.g., QLF-D Biluminator 2+) with consistent settings: aperture f/2.8, exposure time 1/30 sec, ISO 100. Ensure fixed distance (e.g., 30 cm) between camera and sample.
- Image Segmentation: Use proprietary software (e.g., QA2 v2.0.0.12) to separate fluorescence signals. The software splits the captured image into separate green (F520) and red (F630) channel images based on built-in filters.
- Quantitative Analysis:
- Define a Region of Interest (ROI) over the biofilm.
- Measure average pixel intensity for both green (
I_G) and red (I_R) channels within the ROI. - Calculate the Red/Green Ratio (R/G):
R/G = I_R / I_G. - Calculate ΔR (change in red fluorescence) relative to a non-fluorescent reference standard.
- Validation: Correlate R/G values with conventional microbiological assays (CFU counts, porphyrin extraction assays) and confocal laser scanning microscopy (CLSM) with live/dead staining.
Table 2: Expected In Vitro Biofilm Fluorescence Profile Over Time
| Biofilm Age | Dominant Microbiota | Primary Metabolic State | Mean Green Intensity (a.u.) | Mean Red Intensity (a.u.) | Mean R/G Ratio | Correlation with Porpyhrin (nmol/mg) |
|---|---|---|---|---|---|---|
| 24 hours | Streptococci, early colonizers | Aerobic/Glycolytic | 1850 ± 210 | 150 ± 45 | 0.08 ± 0.02 | 0.05 ± 0.01 |
| 72 hours | Mixed, including Actinomyces | Anaerobic pockets | 1120 ± 180 | 680 ± 120 | 0.61 ± 0.08 | 0.42 ± 0.07 |
| 120 hours | Mature, including anaerobes | Predominantly Anaerobic | 750 ± 95 | 1250 ± 210 | 1.67 ± 0.15 | 1.18 ± 0.14 |
Signaling Pathways and Metabolic Origins of Red Fluorescence
Orthodontic Research Application Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Biofilm Fluorescence Research
| Item / Reagent | Function / Rationale | Example Product / Specification |
|---|---|---|
| Hydroxyapatite (HA) Discs | Physiologically relevant substrate for in vitro biofilm growth, mimicking tooth enamel. | Clarkson Chromatography sHA discs, 10mm diameter. |
| Defined Biofilm Media (with Sucrose) | Promotes the formation of cariogenic, polysaccharide-rich biofilms that develop red fluorescence. | McBain medium modified with 1% (w/v) sucrose. |
| Anaerobic Chamber & Gas Packs | Creates the low-oxygen environment essential for porphyrin accumulation and red-fluorescent biofilm maturation. | Coy Laboratory Type B Vinyl Anaerobic Chamber; BD GasPak EZ. |
| Porphyrin Extraction Buffer | For chemical validation of red fluorescence by quantifying extracted porphyrins. | 1M HClO₄:Ethanol (1:1, v/v) extraction buffer. |
| Fluorescence Reference Standards | Calibrates QLF-D devices for consistent, quantitative intensity measurements across sessions. | Non-fluorescent ceramic (ΔR=0) and stable fluorescent plaques. |
| CLSM Live/Dead Stain | Validates biofilm viability and spatial structure correlated with fluorescence signals. | BacLight LIVE/DEAD kit (SYTO9 & Propidium Iodide). |
| Protoporphyrin IX Standard | Quantitative standard for calibrating porphyrin assays and confirming red fluorescence origin. | Sigma-Aldrich Protoporphyrin IX, ≥95% (HPLC). |
| Artificial Saliva | For creating saliva-coated HA discs, providing a pellicle for primary bacterial adhesion. | Mucin-containing formulation (e.g., 2.5 g/L mucin, pH 6.8). |
Fixed orthodontic appliances fundamentally alter the oral ecosystem. Brackets, wires, and ligatures create a complex topography of stagnation areas, drastically impeding natural cleansing mechanisms. This leads to rapid and localized plaque accumulation, biofilm maturation shifts, and an elevated risk of enamel demineralization (white spot lesions, WSLs), the most common iatrogenic complication of orthodontic treatment.
For researchers and drug development professionals, this environment presents a unique, high-fidelity clinical model. The predictable and accelerated plaque dynamics around brackets offer a powerful setting for:
- Evaluating novel anti-plaque/anti-biofilm agents.
- Assessing remineralization therapies.
- Validating new plaque quantification methodologies.
- Studying biofilm ecology under challenging mechanical conditions.
Integrating Quantitative Light-induced Fluorescence-Digital (QLF-D) into orthodontic research protocols provides an objective, longitudinal, and quantitative measure of plaque coverage and bacterial activity, moving beyond subjective indices.
Table 1: Comparative Plaque Metrics in Orthodontic vs. Non-Orthodontic Patients
| Metric | Non-Orthodontic Patients (Control) | Orthodontic Patients (Bracket-Period) | Measurement Tool | Key Study Reference (Search Date: 2026) |
|---|---|---|---|---|
| Plaque Index (Mean) | 0.8 - 1.2 | 1.6 - 2.1 | Modified Silness & Löe Index | Recent systematic reviews |
| Incidence of White Spot Lesions | 2.4% - 11% | 23% - 73% | Visual Examination, QLF, ICDAS | Meta-analyses (2019-2025) |
| Plaque Volumetric Increase | Baseline (1x) | 3x - 5x increase around brackets | 3D intraoral scanning & biofilm analysis | In-vivo studies (2023-2025) |
| Streptococcus mutans Count | ~10^4 - 10^5 CFU/mL saliva | ~10^5 - 10^6 CFU/mL saliva | Microbial culture, qPCR | Longitudinal cohort studies |
| QLF-D Plaque Fluorescence (ΔR_{30}) | Low to moderate red fluorescence | High red fluorescence, particularly at gingival margin of bracket | QLF-D Biluminator | Current clinical trials |
Table 2: QLF-D Output Parameters Relevant to Orthodontic Research
| QLF-D Parameter | Description | Relevance to Orthodontic Research |
|---|---|---|
| ΔR (Red Fluorescence Change) | Quantitative measure of porphyrin-producing bacterial activity. | Primary endpoint for anti-microbial efficacy trials. Correlates with cariogenic biofilm maturity. |
| ΔF (Fluorescence Loss) | Quantitative measure of enamel demineralization. | Key endpoint for remineralization/WSL prevention studies. |
| %Plaque Coverage | Pixel-based calculation of plaque-covered area. | Objective measure of anti-adhesion/mechanical cleansing efficacy. |
| Plaque Severity Distribution | Heat-map visualization of ΔR values across a tooth. | Identifies high-risk zones (gingival, bracket periphery). |
Experimental Protocols
Protocol 1: QLF-D Clinical Imaging for Longitudinal Plaque Assessment in Orthodontic Patients
Objective: To quantitatively monitor plaque accumulation and bacterial activity around orthodontic brackets over time. Materials: QLF-D device (e.g., Inspektor Pro, QLF-D Biluminator), cheek retractors, ADA typodont for calibration, alignment jig (for reproducibility), proprietary analysis software (QA2 v2.0+). Procedure:
- Patient Preparation: Refrain from oral hygiene for 12-24 hours prior to imaging. Rinse with water to remove loose debris.
- Calibration: Perform daily device calibration using a white reference standard.
- Positioning: Use a custom alignment jig to ensure consistent camera-tooth distance and angulation at each visit. Capture the buccal surfaces of target teeth (typically premolars and canines).
- Image Capture: Acquire images under white light, blue light (407 nm) for natural fluorescence (ΔF), and violet light (405 nm) for plaque-induced red fluorescence (ΔR). Ensure the field includes the full bracket, gingival margin, and occlusal edge.
- Analysis: In the software, define the region of interest (ROI) as a 1-mm perimeter around the bracket. The software automatically calculates %Plaque Coverage and mean ΔR within the ROI. A separate ROI can be drawn for enamel demineralization analysis (ΔF).
- Data Export: Export numerical data (ΔR, ΔF, coverage %) for statistical analysis.
Protocol 2: Ex Vivo Bracket-Biofilm Model for High-Throughput Screening
Objective: To screen potential anti-biofilm agents under controlled, orthodontic-relevant conditions. Materials: Extracted human premolars, stainless steel orthodontic brackets, artificial saliva, S. mutans UA159 strain, 24-well culture plates, test compounds (e.g., novel antimicrobial peptides, modified chlorhexidine formulations), confocal laser scanning microscope (CLSM) with live/dead stain (SYTO9/propidium iodide). Procedure:
- Substrate Preparation: Bond brackets to the buccal surface of sterilized, extracted premolars using light-cure orthodontic adhesive.
- Inoculation: Place each tooth in a well. Condition teeth with artificial saliva for 2 hours. Inoculate with S. mutans suspension (~10^7 CFU/mL) in growth medium supplemented with 1% sucrose.
- Biofilm Growth & Treatment: Incubate anaerobically (37°C, 72h), refreshing medium daily. At 48h, apply treatment solutions for 60 seconds (simulating a rinse) or incorporate them into the growth medium.
- Analysis:
- QLF-D: Image ex vivo using a standardized holder.
- Microbiological: Sonicate to harvest biofilm, plate for CFU counts.
- CLSM: Stain biofilm on bracket/tooth interface, image to determine biofilm thickness and viability ratio.
- Endpoint: Correlate QLF-D ΔR values with CFU reduction and CLSM viability data.
Signaling Pathways in Cariogenic Biofilm Formation Around Brackets
Title: Biofilm Pathway Leading to Demineralization
QLF-D Orthodontic Research Workflow
Title: QLF-D Orthodontic Study Protocol
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Orthodontic Plaque Research
| Item | Function & Relevance |
|---|---|
| QLF-D Device (e.g., Inspektor Pro) | Captures quantitative fluorescence images for plaque activity (ΔR) and enamel health (ΔF). The core tool for non-invasive, longitudinal data collection. |
| QA2 v2.0+ Analysis Software | Proprietary software for analyzing QLF-D images. Enables precise ROI selection and automated calculation of key parameters (ΔR, ΔF, % coverage). |
| Custom Alignment Jig (3D printed) | Ensures reproducible camera positioning for serial imaging, critical for longitudinal study validity. |
| Ex Vivo Bracket-Biofilm Model Kit | Standardized components (brackets, adhesive, defined bacterial strains) for controlled, high-throughput screening of agents. |
| Artificial Saliva with Mucin | Maintains tooth/bracket substrate hydration and provides a pellicle mimic for ecologically relevant biofilm growth in vitro. |
| Live/Dead BacLight Bacterial Viability Kit | Used with CLSM to validate QLF-D ΔR readings against direct measures of biofilm cell viability. |
| Standardized Sucrose Challenge Protocol | Defines the frequency and concentration of sucrose pulses in biofilm models, simulating a cariogenic challenge. |
| Digital Caliper & 3D Intraoral Scanner | Provides complementary geometric data (plaque volume, bracket area) for correlative analysis with QLF-D fluorescence data. |
This application note details the quantitative light-induced fluorescence-digital (QLF-D) methodology as the core assessment protocol for a thesis investigating longitudinal plaque dynamics and anti-plaque agent efficacy during fixed orthodontic treatment. The limitations of traditional visual indices like the Plaque Index (PI) and Quigley-Hein Index (QHI) necessitate a shift to objective, sensitive, and digitally archivable systems to generate high-quality data for clinical research and drug development.
Comparative Analysis: Quantitative Metrics
Table 1: Core Comparison of Plaque Assessment Methodologies
| Parameter | Traditional Indices (PI, QHI) | QLF-D (QLF-D Biluminator) |
|---|---|---|
| Assessment Basis | Visual tactile (PI) or visual (QHI) scoring of disclosed plaque. | Quantitative measurement of fluorescence loss (%ΔF) due to porphyrin-producing plaque bacteria. |
| Primary Output | Ordinal score (e.g., 0-3 for PI, 0-5 for QHI). | Continuous, ratio-scale data: %ΔF, Area (pixels²), and Red Fluorescence (R-value) for specific plaque types. |
| Sensitivity | Low to moderate; limited discrimination of early biofilm and small changes post-intervention. | High; detects pre-clinical/mineralized plaque, measures incremental changes in plaque volume and activity. |
| Objectivity & Reproducibility | Subject to inter/intra-examiner variability. Requires calibration but inherently subjective. | High; software-driven analysis (e.g., QA2 v1.27) minimizes human bias. Raw digital data allows re-analysis. |
| Data Capture | Ephemeral; score sheets. No permanent visual record of the actual plaque. | Digital archiving of standardized fluorescence images. Enables retrospective analysis and secondary endpoints. |
| Throughput & Analysis Speed | Fast at chairside but manual data entry required. | Image capture is rapid; software analysis is automated but requires initial region-of-interest selection. |
| Key Limitation | Subjective, coarse grading, unable to quantify plaque biochemistry. | Initial equipment cost. Requires controlled lighting. Calculus can interfere with red fluorescence signals. |
Table 2: Example Sensitivity Data from Comparative Studies
| Study Focus | PI/QHI Outcome | QLF-D Outcome | Implied Advantage |
|---|---|---|---|
| Plaque Growth Inhibition (24h) | QHI: No significant difference between test & control mouthwash (p>0.05). | %ΔF: Significant reduction in plaque fluorescence for test mouthwash (p<0.01). | QLF-D detects sub-visual biochemical changes. |
| Plaque Maturation (72h) | PI: Saturation at max score for both groups, unable to differentiate density. | R-value (Red Fluorescence): Significant increase in mature, porphyrin-rich plaque in control group (p<0.001). | QLF-D quantifies plaque age/bacterial composition. |
| Preventive Agent Efficacy | Visual indices show moderate effect. | Correlation between %ΔF reduction and microbial load (r=0.89, p<0.001). | QLF-D provides continuous data for robust statistical modeling. |
Experimental Protocols
Protocol 1: QLF-D Imaging for Longitudinal Orthodontic Plaque Assessment
Objective: To acquire standardized fluorescence images of plaque around brackets and gingival margins for quantitative analysis over time.
Materials: See "Scientist's Toolkit" below.
Methodology:
- Subject Preparation & Plaque Disclosure: Refrain from oral hygiene for a defined period (e.g., 24h). Rinse with a disclosing solution (e.g., 2-tone) for 10 seconds and then with water for 20 seconds to remove excess dye.
- QLF-D System Calibration: Power on the QLF-D Biluminator and associated software. Perform daily white balance and uniformity calibration using the provided standard reflectance tile.
- Intra-Oral Camera Setup: Mount the sterilized intra-oral camera. Set capture parameters to fixed values: Shutter speed (1/30 sec), Aperture (F8), ISO (200).
- Image Acquisition: a. Position the subject with head stabilized. b. For each target tooth (e.g., maxillary anterior), align the camera perpendicular to the labial surface. c. Ensure the field of view includes the bracket, full crown, and 2-3 mm of gingival margin. d. Instruct the subject to hold breath momentarily to avoid fogging. e. Capture images under white light (for reference) and violet-blue light (405 nm) for fluorescence.
- Image Archiving: Save images in lossless format (e.g., TIFF) with a unique anonymized ID, date, visit, and tooth number.
Protocol 2: Quantitative Analysis of QLF-D Images Using QA2 Software
Objective: To derive objective plaque metrics (%ΔF, Area, R-value) from captured fluorescence images.
Methodology:
- Software Initialization: Launch QA2 software and load the fluorescence image of the target tooth.
- Region of Interest (ROI) Definition: Using the "Tooth ROI" tool, meticulously trace the anatomical crown contour, excluding the gingiva and bracket. For specific analysis, sub-ROIs (e.g., gingival 1/3, perimeter around bracket) can be defined.
- Reference Selection: Use the "Auto Loss" tool to select a sound, non-fluorescent area of enamel as the reference fluorescence level (F0).
- Plaque Threshold Setting: Set the fluorescence loss threshold (typically ΔF = -5% to -10%) to differentiate plaque from sound enamel. The software will calculate the area with fluorescence below this threshold.
- Data Extraction: a. %ΔF: The average fluorescence loss within the defined plaque area. b. Area: The size of the plaque area in pixels (convertible to mm²). c. R-value: The ratio of red-to-green fluorescence intensity, calculated in specific "Red" analysis mode, indicating mature plaque.
- Data Export: Export all numerical data to CSV/Excel for statistical analysis.
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Function in QLF-D Protocol |
|---|---|
| QLF-D Biluminator (Inspektor Pro) | Integrated light source (405 nm) & camera system for standardized fluorescence image capture. |
| QA2 Image Analysis Software (v1.27+) | Proprietary software for analyzing fluorescence images, calculating %ΔF, Area, and R-value. |
| 2-Tone Disclosing Solution | Stains mature (blue) and new (pink) plaque, aiding visual reference and ROI selection, though not required for QLF signal. |
| Calibration Standard Tile | Provides a uniform white reflectance reference for daily system calibration and white balancing. |
| Sterilizable Intra-Oral Camera Tips | Ensures cross-infection control during image capture. |
| Chin/Head Rest Stabilizer | Minimizes subject movement, ensuring consistent image framing and focus across longitudinal visits. |
| Data Archiving Software (e.g., XDR) | Secured database for managing thousands of anonymized QLF-D images and linked metadata. |
Visualizations
Diagram 1: QLF-D vs Traditional Indices Workflow Comparison
Diagram 2: QLF-D Plaque Quantification Logic Pathway
Diagram 3: Thesis Clinical Assessment Protocol Flow
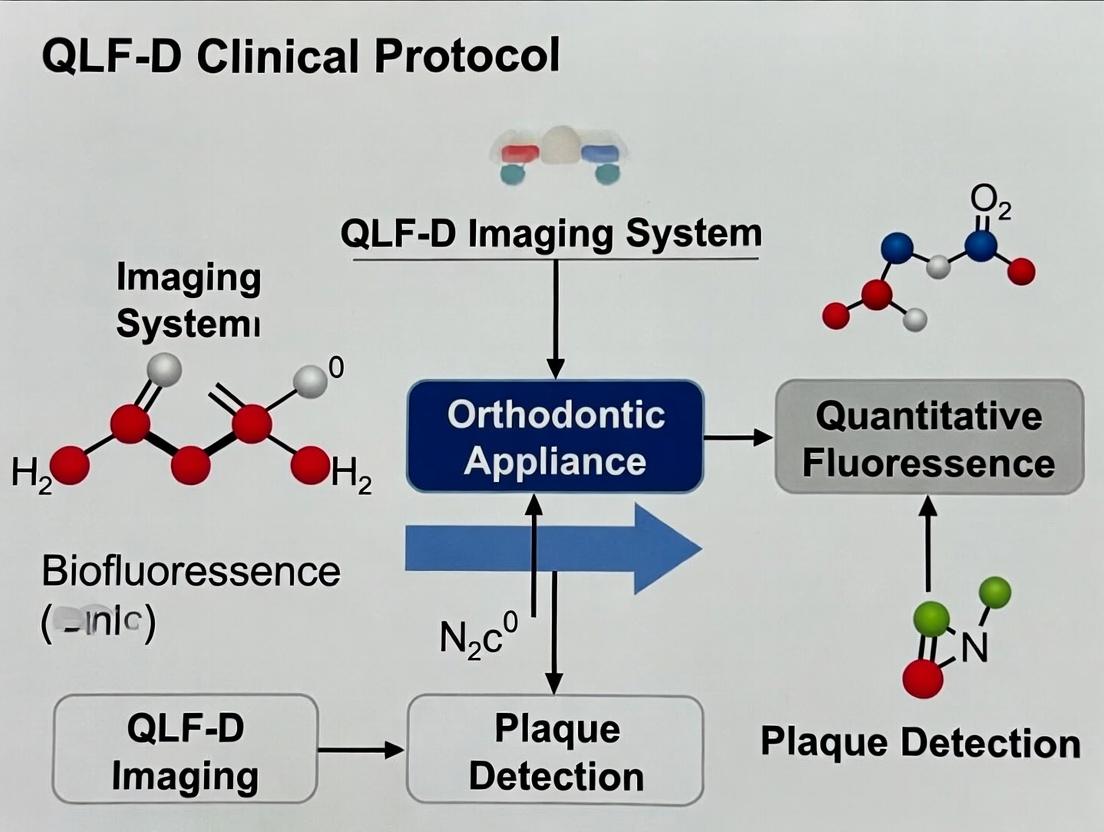
Within the broader thesis on establishing a Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocol for plaque assessment during orthodontic research, this document details the application and measurement of three key parameters: ΔR (Red Fluorescence change), ΔF (Fluorescence loss), and Lesion Area. These parameters are critical for quantifying plaque biofilm activity, acidogenicity, and maturity, which are exacerbated by fixed orthodontic appliances. Their measurement enables the objective evaluation of preventive interventions and oral chemotherapeutics in longitudinal clinical trials.
Quantitative Parameters: Definitions and Biological Significance
ΔR: Red Fluorescence Intensity Change
ΔR quantifies the increase in red fluorescence emitted by dental plaque when illuminated with blue-violet light (≈405 nm). This signal is primarily attributed to porphyrins and related compounds produced by specific cariogenic bacteria (e.g., Porphyromonas gingivalis, Prevotella spp.).
Biological Significance: Serves as a direct biomarker for mature, metabolically active, and often pathogenic anaerobic bacterial populations within the biofilm. A higher ΔR correlates with increased plaque maturity and caries risk.
ΔF: Fluorescence Loss
ΔF represents the percentage loss of green fluorescence from tooth enamel compared to a sound reference area. In QLF-D, healthy enamel emits strong green fluorescence under blue light; demineralization (early caries) causes a quantifiable decrease.
Biological Significance: ΔF is the primary metric for quantifying early enamel demineralization (white spot lesions) adjacent to orthodontic brackets, a direct consequence of prolonged plaque biofilm activity.
Lesion Area (mm²)
Lesion Area is the two-dimensional size of the demineralized region on the enamel surface, as demarcated by the fluorescence loss boundary (typically using a ΔF threshold of -5%).
Biological Significance: Provides a spatial measure of demineralization extent. Combined with ΔF, it calculates the total "Fluorescence Loss Volume," offering a comprehensive assessment of demineralization severity.
Table 1: Summary of Key Measurable Parameters
| Parameter | Symbol | Unit | Measurement Source | Biological/Clinical Significance |
|---|---|---|---|---|
| Red Fluorescence Change | ΔR | ΔR (unitless) or % | Plaque Biofilm | Biomarker for mature, cariogenic bacterial activity. |
| Fluorescence Loss | ΔF | % | Tooth Enamel | Quantifies early enamel demineralization (white spots). |
| Lesion Area | Area | mm² | Tooth Enamel | Spatial extent of demineralization. |
Detailed Experimental Protocols
Protocol for Concurrent ΔR and ΔF Assessment in Orthodontic Patients
Aim: To longitudinally monitor plaque bioactivity and enamel demineralization around brackets.
Materials:
- QLF-D Biluminator 2+ (Inspektor Research Systems) or similar validated device.
- Calibration block.
- Metal retractors, cheek retractors.
- Intra-oral camera stand for stabilization.
- Image analysis software (QA2 v.1.2 or later).
Procedure:
- Subject Preparation & Imaging:
- Schedule visit post-prophylaxis (baseline) and at defined intervals (e.g., 4, 8, 12 weeks).
- Use retractors to fully expose the dental arch with fixed appliances.
- Stabilize the QLF-D handpiece using the camera stand. Position perpendicular to tooth surface.
- Capture images: a) White-light reflectance image. b) Fluorescence image (405 nm excitation, filters for green/red emission).
Image Analysis for ΔR (Plaque):
- In software, load the fluorescence image.
- Select a standardized region of interest (ROI) on the plaque adjacent to the bracket gingival margin.
- Use the software's "Red Fluorescence" analysis tool. The software calculates the average red/green ratio (R) within the ROI.
- ΔR is calculated as the difference in R value from the baseline plaque-free image (or a sound reference area).
ΔR = R_sample - R_reference.
Image Analysis for ΔF and Lesion Area (Enamel):
- Select an ROI on the enamel surface bordering the bracket.
- The software reconstructs the predicted fluorescence of sound enamel based on surrounding pixel intensities.
- Set the threshold for demineralization at a fluorescence loss of -5%.
- The software automatically calculates:
- ΔF: The average percentage fluorescence loss within the demarcated lesion.
- Lesion Area: The area (mm²) of the region exceeding the -5% threshold.
Data Management:
- Record ΔR, ΔF (%), and Lesion Area (mm²) for each site per time point.
- Calculate ΔF × Area (fluorescence loss volume) for comprehensive analysis.
Protocol forIn VitroPlaque Maturation and Drug Efficacy Screening
Aim: To correlate ΔR with plaque biofilm maturity and test anti-biofilm agents.
Materials:
- Microbial strain consortium (e.g., S. mutans, L. casei, A. naeslundii, P. gingivalis).
- Hydroxyapatite discs (HA discs) simulating tooth enamel.
- Artificial saliva and growth media (TYCSB).
- Anaerobic workstation.
- QLF-D system adapted for in vitro use or spectrophotometer with 405 nm excitation.
Procedure:
- Biofilm Growth:
- Sterilize HA discs in an ultraviolet chamber.
- Inoculate discs in 24-well plates with the microbial consortium. Culture anaerobically (37°C, 72 hrs+), with daily media changes to simulate plaque maturation.
Treatment Application (For drug screening):
- At 72 hrs, apply experimental antimicrobial/antiplaque solutions (e.g., CHX, novel compounds) to test groups. Use PBS for negative control.
- Incubate for determined exposure time (e.g., 1 min, 2x daily).
ΔR Measurement In Vitro:
- Rinse discs gently to remove non-adherent cells.
- Place disc in a dark imaging chamber. Acquire fluorescence image with QLF-D or spectral device.
- Analyze mean red fluorescence intensity from the disc image. Report as ΔR relative to an untreated, sterile HA disc control.
Validation Assays (Correlative):
- Perform CFU counting (viability) and Confocal Laser Scanning Microscopy (CLSM) with LIVE/DEAD staining on parallel samples to validate ΔR correlations with bacterial load/viability.
Table 2: Example Experimental Data from a 4-Week Orthodontic Study
| Patient ID | Tooth Site | Baseline ΔR | Week 4 ΔR | Baseline ΔF% | Week 4 ΔF% | Week 4 Lesion Area (mm²) |
|---|---|---|---|---|---|---|
| 001 | 11 Facial | 0.15 | 0.42 | -2.1 | -8.7 | 0.85 |
| 001 | 36 Buccal | 0.18 | 0.38 | -1.8 | -6.5 | 0.52 |
| 002 | 22 Facial | 0.12 | 0.61 | -0.9 | -12.4 | 1.20 |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for QLF-D Plaque & Demineralization Research
| Item | Function in Research |
|---|---|
| QLF-D Clinical Imaging System (e.g., Inspektor Pro) | Core device for standardized capture of fluorescence (green/red) and white-light images in vivo. |
| QA2 Image Analysis Software | Proprietary software for quantitative analysis of ΔF, ΔR, and Lesion Area from captured images. |
| Hydroxyapatite (HA) Discs | Synthetic enamel substrate for growing standardized in vitro plaque biofilms for drug testing. |
| Anaerobic Chamber (Workstation) | Essential for culturing the anaerobic species responsible for red fluorescence in mature plaque. |
| Bacterial Strain Consortium | Defined microbial community (including porphyrin producers) to simulate cariogenic plaque. |
| Confocal Laser Scanning Microscope (CLSM) | Gold-standard for validating biofilm architecture and viability correlating with ΔR measurements. |
| Fluorescent Vital Stains (e.g., SYTO 9, PI) | Used with CLSM for quantitative viability analysis (Live/Dead) of biofilms. |
Visualization Diagrams
Title: Pathway from Orthodontic Plaque to QLF-D Parameters
Title: QLF-D Clinical Trial Workflow for Orthodontics
A rigorous pre-treatment assessment is foundational for clinical trials in orthodontic research, particularly those investigating interventions for white spot lesions (WSLs) or enamel demineralization. Quantitative Light-Induced Fluorescence-Digital (QLF-D) technology provides an objective, non-invasive method for quantifying early enamel lesions. This protocol details the establishment of baseline assessments and patient stratification criteria using QLF-D within the context of orthodontic clinical trials, ensuring reproducible cohorts for evaluating preventive or therapeutic agents.
Application Notes: The Role of QLF-D in Baseline Assessment
QLF-D measures the loss of autofluorescence due to bacterial metabolites and enamel porosity. Baseline QLF-D parameters are critical for:
- Primary Outcome Definition: Establishing the initial severity of WSLs (ΔF, Area).
- Patient Stratification: Creating homogenous subgroups to reduce outcome variability.
- Monitoring Progression/Regression: Providing a reference for calculating ΔQ (change in lesion volume) over time.
Core Assessment and Stratification Protocol
Pre-Treatment Screening and Inclusion Workflow
Diagram Title: Patient Screening and Stratification Workflow for Orthodontic Trials
Standardized QLF-D Image Acquisition Protocol
Objective: To capture reproducible, quantitative baseline fluorescence images of labial enamel surfaces. Materials: See Research Reagent Solutions table. Procedure:
- Patient Preparation: Clean teeth with water and non-fluoridated pumice for 10 seconds per tooth. Rise thoroughly for 30 seconds.
- Isolation & Drying: Isolate the arch with cheek retractors. Air-dry tooth surfaces for 10 seconds using a standardized dental syringe (air pressure ~0.3 MPa).
- Image Acquisition:
- Position the QLF-D handpiece perpendicular to the tooth surface at a fixed distance (as per manufacturer).
- Ensure the imaging window covers the target teeth (typically canines to canines).
- Capture images under autofluorescence mode. Save images in proprietary format for analysis.
- Quality Control: Check images for focus, dryness, and absence of artifacts (e.g., saliva, plaque droplets).
Key Baseline Metrics and Stratification Criteria
The following quantitative data, derived from the QLF-D analysis software, forms the basis for patient stratification.
Table 1: Key QLF-D Baseline Metrics and Recommended Stratification Thresholds
| Metric | Definition | Measurement Unit | Stratification Thresholds for Orthodontic Trials |
|---|---|---|---|
| ΔF | Average percentage loss of fluorescence within the lesion area. | % | High Risk: ΔF ≤ -5% Moderate Risk: -5% < ΔF ≤ 0% |
| Lesion Area | Two-dimensional area of the lesion with fluorescence loss below threshold. | mm² | High Risk: Area ≥ 3.0 mm² Moderate Risk: Area < 3.0 mm² |
| ΔQ (Baseline) | Integrated fluorescence loss (ΔF × Area). Represents lesion "volume." | %·mm² | High Risk: ΔQ ≤ -15 %·mm² Moderate Risk: ΔQ > -15 %·mm² |
Table 2: Example Stratification Matrix for Trial Randomization
| Patient ID | Baseline ΔF (%) | Baseline Area (mm²) | Baseline ΔQ (%·mm²) | Stratified Cohort |
|---|---|---|---|---|
| P-001 | -7.2 | 4.1 | -29.5 | A: High Risk |
| P-002 | -3.1 | 1.8 | -5.6 | B: Moderate Risk |
| P-003 | -9.5 | 5.5 | -52.3 | A: High Risk |
| P-004 | -1.5 | 0.9 | -1.4 | B: Moderate Risk |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for QLF-D Baseline Assessment Protocol
| Item | Function/Description | Example Product/Specification |
|---|---|---|
| QLF-D Imaging System | Device that induces and captures autofluorescence. Enables quantification of enamel health. | Inspektor Pro with QLF-D Biluminator 2+ |
| Analysis Software | Software to analyze QLF-D images, calculate ΔF, Area, and ΔQ metrics. | QA2 v2.0+ Software |
| Non-Fluoridated Prophy Paste | For standardized cleaning without altering baseline enamel fluorescence. | Nupro Fine or coarse pumice. |
| Chest Retractors | To provide consistent intra-oral access and field isolation. | Disposable plastic cheek retractors. |
| Calibrated Drying Syringe | Provides consistent air pressure for surface drying, a critical pre-imaging step. | 3-way dental syringe, pressure ~0.3 MPa. |
| Calibration Standard | A fluorescent standard for periodic system calibration to ensure longitudinal data consistency. | Manufacturer-supplied calibration tile. |
| Digital Intra-Oral Camera | For capturing standard clinical photographs to correlate with QLF-D findings. | EOS DSLR with macro lens and ring flash. |
Integrated Pathway: From Baseline to Outcome Assessment
Diagram Title: Trial Design Pathway from Baseline to Analysis
Step-by-Step QLF-D Clinical Protocol: Standardization for Reproducible Research Data
This document establishes the standardized pre-procedural protocol for Quantitative Light-induced Fluorescence-Digital (QLF-D) imaging within a longitudinal research study assessing plaque dynamics during fixed orthodontic treatment. Consistent pre-acquisition setup is critical for minimizing inter- and intra-session variability, ensuring data comparability, and validating the quantification of plaque fluorescence (ΔR) and bacterial metabolic activity (ΔF). This protocol is a foundational component of a broader thesis on objective plaque indices for orthodontic research.
Calibration Protocol
White Balance & Color Calibration
- Purpose: To standardize color representation and white balance across all imaging sessions, correcting for ambient light contamination and camera sensor drift.
- Methodology:
- Use a certified, neutral white balance reference card (e.g., X-Rite ColorChecker Classic Mini).
- Under standardized clinic lighting (see Section 3), position the QLF-D handpiece at the typical working distance (e.g., 10 mm) from the card, filling the frame.
- Capture an image in the device's software.
- Execute the "Set White Balance" function within the proprietary analysis software (e.g., QA2 v2.0+).
- Validate by capturing a second image; the RGB values of the white patch should read within ±5 units of neutral (R=G=B).
Fluorescence Intensity Calibration
- Purpose: To ensure consistent and accurate quantification of fluorescence loss (ΔF) over time and between subjects.
- Methodology:
- Use a stable, non-bleaching fluorescent calibration standard (e.g., a resin block doped with a known concentration of fluorophore like Rhodamine B).
- Prior to each clinical imaging day, image the standard under identical exposure settings (shutter speed, aperture, ISO/gain) used for patient imaging.
- In the analysis software, measure the average fluorescence intensity (ΔF) of a defined Region of Interest (ROI) on the standard.
- Record the value. A deviation of >5% from the established baseline ΔF value for the standard necessitates investigation of the light source (LED output) and camera sensor before proceeding.
Table 1: Calibration Schedule and Tolerances
| Calibration Type | Frequency | Standard Used | Acceptance Tolerance | Corrective Action if Failed |
|---|---|---|---|---|
| White Balance | Before each patient session | Neutral white reflectance card | RGB values ±5 from neutral | Re-perform in controlled light; check light source color temp. |
| Fluorescence Intensity | Daily, before first patient | Certified fluorescent standard | ΔF value within ±5% of baseline | Inspect/clean lens; verify LED function; service device. |
| Spatial Scale/Distortion | Quarterly | Ruler/grid target at known distance | Measured length within ±2% of actual | Recalibrate spatial mapping in software. |
Environmental Controls
Ambient Light Control
- Protocol: All QLF-D imaging must be conducted in a room with blackout curtains or no windows. Overhead fluorescent/LED lights must be switched off. The only permissible light sources are the QLF-D device's own LEDs and, if necessary, a dedicated, dim, 405-nm safe examination light for operator positioning.
- Validation: Use a digital lux meter. Measured ambient light at the subject's mouth position must be < 5 lux.
Temperature and Humidity
- While QLF-D is relatively robust, extreme conditions can affect electronic performance and patient comfort.
- Protocol: Maintain room temperature between 20-24°C (68-75°F) and relative humidity between 30-60%.
- Rationale: Stabilizes camera sensor noise and prevents lens fogging.
Patient Preparation Protocol
Pre-Visit Instructions
- Patients are instructed to refrain from using any whitening toothpaste, mouthwash, or chewing gum for 48 hours prior to the research visit.
- They are asked to avoid consuming food or beverages (except water) for at least 1 hour before the appointment to reduce transient staining.
In-Clinic Preparation
- Rinsing: Patient rinses thoroughly with water for 30 seconds to remove loose debris.
- Drying: The tooth surfaces to be imaged are isolated with cotton rolls and gently air-dried using a triple syringe (air only) for 10 seconds per quadrant. Critical: Over-drying must be avoided as it can alter fluorescence.
- Disclosure (Optional, Protocol-Dependent): Only if the research protocol includes comparative assessment with disclosed plaque. A non-fluorescing disclosing agent (e.g., erythrosine) may be applied sparingly with a microbrush, followed by a second gentle water rinse and drying. Note: This must be performed after QLF-D imaging if the native fluorescence of undisclosed plaque is the primary endpoint.
Table 2: Patient Preparation Steps and Rationale
| Step | Procedure Detail | Time | Scientific Rationale |
|---|---|---|---|
| 1. Pre-Visit Abstention | No whitening agents for 48h. | 48 hours | Prevents chemical alteration of endogenous fluorophores (porphyrins) in plaque. |
| 2. Fasting | No food/drink (except water) 1h prior. | 1 hour | Minimizes interference from exogenous food pigments and recent sugar challenge. |
| 3. Rinsing | Vigorous water rinse. | 30 seconds | Removes non-adherent materia alba without disturbing biofilm. |
| 4. Isolation & Drying | Cotton rolls + air syringe. | 10 sec/quadrant | Reduces saliva-induced light scattering; dry surface is optimal for QLF analysis. |
| 5. Disclosure (Optional) | Apply & rinse non-fluorescent agent. | Protocol-specific | Allows for visual plaque index (e.g., Modified Quigley-Hein) correlation with QLF-D data. |
Experimental Workflow Diagram
Title: QLF-D Pre-Procedural and Imaging Workflow
The Scientist's Toolkit: Key Research Reagent Solutions & Materials
Table 3: Essential Research Materials for QLF-D Plaque Assessment
| Item | Function/Application | Example Product/Specification |
|---|---|---|
| QLF-D Clinical System | Image acquisition hardware. | Inspektor Pro QLF-D Clinic System with QA2 software. |
| Neutral White Balance Card | Calibrates camera white balance for true color representation. | X-Rite ColorChecker Classic Mini. |
| Fluorescent Calibration Standard | Provides a stable reference for quantitative fluorescence (ΔF) validation. | Custom Rhodamine B-doped acrylic block (λex ~405nm, λem ~590nm). |
| Digital Lux Meter | Quantifies and validates ambient light levels pre-imaging. | Lutron LX-101A or equivalent (range 0.1 - 200,000 lux). |
| Triple Syringe (Air Only) | Provides controlled, moisture-free air for drying tooth surfaces. | Standard dental unit syringe, air line filtered. |
| Non-Fluorescing Disclosing Agent | Visually stains plaque for comparative indices without interfering with native QLF signal. | Erythrosine-based tablet or solution (e.g., Cetylite). |
| Cotton Rolls & Holders | Isolate arches and control saliva during drying and imaging. | Standard dental cellulose cotton rolls. |
| Retractors (Cheek/Lip) | Improve access and field of view for posterior teeth. | Sterile, single-use plastic retractors. |
| Reference Ruler/Grid | For spatial calibration and scale verification in images. | 2mm grid sticker or hand-held scale. |
Within the broader thesis on establishing a clinical protocol for Quantitative Light-induced Fluorescence-Digital (QLF-D) plaque assessment during orthodontic treatment, standardized intraoral imaging is foundational. Reliable, reproducible image capture is critical for longitudinal quantification of plaque fluorescence intensity and area, especially around brackets and wires. This document details the protocols for full-arch and bracket-specific captures to ensure data consistency across research sites and time points.
Core Imaging Parameters: Angles, FOV, and Lighting
Standardization minimizes variability in fluorescence signal capture caused by changes in camera-to-subject distance, angle, and incident light.
Table 1: Standardized Imaging Protocol Parameters
| Parameter | Full-Arch Capture | Bracket-Specific Capture | Rationale |
|---|---|---|---|
| Camera Type | Intraoral camera with QLF-D filter set (e.g., Inspektor Pro, QLF-D Clinic). | Same as Full-Arch. | Ensures consistent excitation (405 nm) and emission (520-550 nm) wavelength capture. |
| Field of View (FOV) | Sufficient to capture entire maxillary or mandibular arch from second molar to second molar. | Isolate 2-3 adjacent teeth with brackets; FOV approx. 30 x 23 mm. | Full-arch provides overall plaque distribution. Bracket-specific enables pixel-level analysis of plaque accumulation zones. |
| Working Distance | Fixed at 5 mm from the incisal edges/occlusal surfaces using a sterile spacer. | Fixed at 5 mm from the labial/buccal surface using a spacer. | Standardizes magnification and minimizes focus-induced intensity variance. |
| Angulation (Camera) | Perpendicular to the occlusal plane for maxillary arch; perpendicular to the labial surface of central incisors for frontal. | Perpendicular to the labial/buccal surface of the tooth of interest. | Orthogonal capture minimizes geometric distortion and ensures even lighting. |
| Angulation (Light Source) | Co-axial with lens axis (integrated ring flash). | Co-axial with lens axis (integrated ring flash). | Eliminates shadows and provides even illumination across FOV. |
| Illumination Intensity | Fixed, pre-calibrated intensity level (e.g., 80% of max output). Document setting. | Fixed, identical to full-arch setting. | Critical for reproducible fluorescence intensity values. |
| Ambient Light Control | Dental operatory lights off. Use blackout curtains if necessary. | Complete darkness except for camera light. | Eliminates contamination from ambient white light. |
| Image Resolution | Minimum 1920 x 1080 pixels (Full HD). | Minimum 1920 x 1080 pixels (Full HD). | Sufficient detail for plaque segmentation software analysis. |
Detailed Experimental Protocols
Protocol 3.1: Pre-Imaging Calibration & Setup
- Device Warm-up: Power on QLF-D system 15 minutes prior to imaging for LED stability.
- White Balance & Intensity Calibration: Using a standardized white reference tile provided with the system, perform white balance and confirm illumination intensity per manufacturer instructions. Record calibration values.
- Patient Preparation: Instruct patient to rinse mouth with water to remove loose debris. Do not use disclosing agents. Retract cheeks and lips using sterile, single-use plastic retractors.
Protocol 3.2: Full-Arch Image Capture Sequence
- Position patient upright with head stabilized in headrest. Align Frankfort plane parallel to the floor.
- Maxillary Arch: Position camera perpendicular to the occlusal plane. Use sterile 5mm spacer. Capture image ensuring all teeth from second molar to second molar are in FOV.
- Mandibular Arch: Reposition camera perpendicular to the occlusal plane. Capture using same spacer distance.
- Labial View (Frontal): Position camera perpendicular to the labial surface of maxillary central incisors. Capture with lips fully retracted.
- Save images in uncompressed format (e.g., TIFF) with filename:
[PatientID]_[Arch(U/L/F)]_[Date_YYYYMMDD].tiff
Protocol 3.3: Bracket-Specific Image Capture Sequence
- Identify target teeth based on study design (e.g., maxillary right canine to first premolar).
- Use a single-tooth, sterile retractor to isolate the area.
- Position camera perpendicular to the labial/buccal surface of the target tooth, ensuring the bracket is centered in the FOV. Use 5mm spacer.
- Capture image ensuring sharp focus on the bracket wings and gingival margin.
- Save images as:
[PatientID]_[ToothFDInotation]_[Date].tiff
Diagram: QLF-D Orthodontic Plaque Assessment Workflow
Diagram Title: Standardized QLF-D Imaging & Analysis Pipeline for Orthodontics
The Scientist's Toolkit: Essential Research Reagent Solutions & Materials
Table 2: Key Materials for QLF-D Orthodontic Plaque Research
| Item | Function in Protocol | Specification Notes |
|---|---|---|
| QLF-D Imaging System | Captures autofluorescence of dental plaque. | Must include 405 nm excitation filter, 520-550 nm emission filter, and proprietary software (e.g., Inspektor Pro system). |
| Sterle Single-Use Spacers | Standardizes working distance (WD). | 5mm thickness, autoclavable or disposable. Critical for reproducibility. |
| Plastic Lip & Cheek Retractors | Provides consistent soft tissue retraction. | Single-use, transparent to avoid interference. |
| Calibration Reference Tile | Standardizes white balance and light intensity. | Manufacturer-provided, ceramic white reference. Calibrate daily. |
| Head Stabilization System | Standardizes patient head position. | Dental chair with adjustable headrest; consider external head stabilizer for longitudinal studies. |
| Data Management Software | Handles image metadata and storage. | Should support DICOM or detailed tagging (Patient ID, Date, Tooth #, Arch, Camera Settings). |
| Plaque Analysis Software | Quantifies plaque fluorescence parameters. | QLF-D proprietary software (e.g., QA2) for calculating ΔF (fluorescence loss) and plaque area. |
| Blackout Curtains / Enclosure | Controls ambient light contamination. | Essential for eliminating variable ambient light, especially in multi-chair clinics. |
This application note details critical methodologies for defining Regions of Interest (ROIs) within the broader thesis on establishing a Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocol for longitudinal plaque assessment in orthodontic research. Accurate, reproducible segmentation of the bracket periphery, gingival margins, and interproximal areas is foundational for quantifying plaque fluorescence dynamics, evaluating anti-plaque agents, and correlating biofilm accumulation with clinical outcomes such as enamel demineralization and gingival inflammation.
ROI Definitions and Clinical Significance
Table 1: Defined ROIs and Their Clinical Significance in Orthodontic Plaque Assessment
| ROI | Anatomical/Appliance Definition | Primary Role in Plaque Assessment | Associated Risk |
|---|---|---|---|
| Bracket Periphery | 0.5-1.0 mm annulus around bracket base adhesive margin. | Quantifies "critical zone" plaque leading to white spot lesions. | Enamel demineralization. |
| Free Gingival Margin | Coronal 1-2 mm of gingival tissue following scalloped contour. | Assesses plaque at gingivitis initiation site. | Marginal gingivitis. |
| Interproximal Area | Clinical contact point to crest of gingival papilla (mesial/distal). | Evaluates plaque in cleansable but risk-prone area. | Interproximal caries, papillary gingivitis. |
Segmentation Strategies and Protocols
Image Acquisition Pre-Processing for ROI Definition
Protocol: Standardized QLF-D Image Capture
- Equipment: QLF-D Biluminator 2+ (Inspektor Research Systems).
- Intraoral Conditions: Professional prophylaxis 48h prior, no brushing for 24h prior to imaging.
- Image Capture: Use lip/cheek retractors. Align camera sensor parallel to tooth surface. Capture under 405 nm violet-blue light with yellow filter (F#2.2, 1/30 sec, ISO 400). Include a standard gray reference card (20% reflectance) in frame for calibration.
- Export: Save images in uncompressed TIFF format (3872 x 2592 pixels).
Semi-Automated Segmentation Workflow
A hybrid approach combining manual landmarking with algorithmic processing yields optimal reproducibility.
Protocol: Manual Landmarking for Algorithm Initialization
- Software: Import TIFF into ImageJ (Fiji) with custom macro.
- Bracket Landmarking: Using the "Point" tool, mark the geometric center of each orthodontic bracket.
- Gingival Margin Landmarking: Using the "Polygon" tool, trace the scalloped cementoenamel junction (CEJ) or, if not visible, the coronal-most free gingival margin across the sextant.
- Interproximal Area Delineation: Mark the clinical contact point and the apical-most extent of the gingival papilla for each interproximal space.
Protocol: Automated ROI Generation via Thresholding & Distance Mapping
- Tooth/Bracket Segmentation: Apply Hue-Saturation-Value (HSV) thresholding to isolate tooth and bracket structures from background.
- Bracket Periphery ROI:
- Create a binary mask from the bracket center point using a region-growing algorithm limited by bracket edge detection.
- Apply a 1-pixel dilation (approx. 0.05 mm) to this mask to define the inner boundary.
- Apply a 20-pixel dilation (approx. 1.0 mm) to create the outer boundary.
- The ROI is the annulus between the two boundaries.
- Gingival Margin ROI:
- Using the traced gingival line as a guide, generate a 30-pixel wide (approx. 1.5 mm) ribbon ROI extending coronally from the line.
- Interproximal ROI:
- Define a polygon connecting the contact point, papilla apex, and the mid-points of the adjacent gingival margin lines.
- Restrict the ROI to the area within the polygon that is part of the tooth surface mask.
Diagram: Segmentation workflow for QLF-D ROIs.
Validation Protocol: Agreement Analysis
Protocol: Comparing Semi-Automated vs. Fully Manual Segmentation
- Operators: Two trained examiners perform fully manual segmentation of all three ROIs on a test set of 50 images.
- Comparison: Compare manual outlines to semi-automated outputs using Dice-Similarity Coefficient (DSC) and Bland-Altman analysis for resultant ΔR30 (red fluorescence) values.
- Acceptance Criterion: DSC >0.85 and mean difference in ΔR30 <5% for all ROI types.
Table 2: Validation Metrics for Semi-Automated Segmentation (n=50 images)
| ROI Type | Mean Dice Score (±SD) | Mean ΔR30 Difference (±SD) | Intraclass Correlation (ICC) |
|---|---|---|---|
| Bracket Periphery | 0.89 (±0.04) | 2.1% (±1.8%) | 0.96 |
| Gingival Margin | 0.87 (±0.05) | 3.4% (±2.5%) | 0.93 |
| Interproximal Area | 0.84 (±0.06) | 4.2% (±3.1%) | 0.91 |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for QLF-D Plaque Assessment Research
| Item | Function in Protocol | Example/Specification |
|---|---|---|
| QLF-D Biluminator 2+ | Provides standardized 405 nm excitation & image capture. | Inspektor Research Systems. |
| Calibration Reference Card | Ensures consistency in white balance and light intensity across sessions. | 20% diffuse gray reflectance. |
| Image Analysis Software | Platform for semi-automated ROI segmentation and ΔF/ΔR calculation. | ImageJ (Fiji) with custom macros; QA2 v1.27 (Inspektor). |
| Fluorescent Plaque Disclosing Gel | Positive control for validating plaque detection sensitivity. | Contains phloxine B or fluorescein. |
| Anti-Plaque Agent (Test) | Intervention to evaluate efficacy on plaque dynamics within ROIs. | e.g., 0.05% CPC mouthrinse, probiotic formulations. |
| Digital Stylus & Graphic Tablet | Enables precise manual landmarking and trace adjustments. | Wacom Intuos Pro. |
Diagram: ROI definition rationale within thesis aim.
The precise definition of the bracket periphery, gingival margin, and interproximal ROIs using the described semi-automated segmentation strategies is critical for generating high-fidelity, quantitative plaque fluorescence data. This standardized approach, embedded within the larger QLF-D clinical protocol, enables robust longitudinal assessment of plaque accumulation patterns and therapeutic interventions in orthodontic patients, directly contributing to evidence-based preventive care strategies.
Quantitative Light-induced Fluorescence Digital (QLF-D) is a validated, non-invasive imaging technology used for the longitudinal assessment of dental plaque during orthodontic treatment. Its application in clinical research protocols allows for the objective quantification of plaque coverage and fluorescence characteristics, which are critical for evaluating oral hygiene efficacy, orthodontic material interactions, and the impact of therapeutic interventions. This application note details the software workflows for image analysis, comparing automated and manual plaque detection methodologies, and standardizing data export for robust statistical analysis within a thesis framework.
Core Software Analysis Workflow
The fundamental workflow for QLF-D image analysis involves sequential steps from image acquisition to data interpretation. The process must be consistent to ensure reproducibility in longitudinal orthodontic studies.
Diagram Title: QLF-D Image Analysis Core Workflow
Automated vs. Manual Plaque Detection: Protocols & Comparison
Detailed Protocol for Automated Detection
Principle: Software algorithms (e.g., in QA2 v.1.26 or custom ImageJ/Python scripts) identify plaque based on fluorescence loss (ΔF) and red fluorescence thresholds.
Materials & Protocol:
- Load Calibrated Image: Import the standardized QLF-D image (
.tiffformat preferred) into analysis software. - Define Tooth Region of Interest (ROI): Manually or via automated tooth segmentation, select the area for analysis (e.g., vestibular surfaces of teeth 13-23). Exclude gingiva and brackets if assessing plaque on enamel.
- Set Detection Thresholds:
- ΔF Threshold: Typically set at -5% to -10% to distinguish demineralized/enhanced plaque areas from sound enamel.
- Red/Green Ratio (R/G): Set a threshold (e.g., R/G > 0.6) to identify mature plaque exhibiting red fluorescence from porphyrins.
- Algorithm Execution: Run the plaque detection function. The software classifies each pixel within the ROI as "plaque" or "non-plaque" based on the dual thresholds.
- Output Generation: Software calculates and displays total plaque coverage (%) and red plaque coverage (%).
Detailed Protocol for Manual Detection
Principle: A trained researcher visually identifies and delineates plaque margins on the digital image.
Materials & Protocol:
- Blinding & Calibration: The investigator is blinded to subject identity and time point. Calibration sessions using a standard set of images are conducted to ensure inter-/intra-rater reliability (Kappa >0.8).
- ROI Definition: As per Step 2 in 3.1.
- Manual Tracin: Using the software's tracing tool (e.g., polygon or freehand), meticulously outline all visible plaque areas within the ROI. Different layers or colors can be used to distinguish between total plaque and red fluorescent plaque.
- Area Calculation: The software calculates the pixel area of all tracings. Plaque coverage is derived as (Plaque Pixel Area / Total ROI Pixel Area) * 100.
Quantitative Comparison of Methods
Table 1: Comparison of Automated vs. Manual QLF-D Plaque Detection
| Parameter | Automated Detection | Manual Detection | Notes for Orthodontic Context |
|---|---|---|---|
| Analysis Time (per image) | ~1-2 minutes | ~5-10 minutes | Automated offers significant efficiency for large cohort studies. |
| Inter-Method Reliability (Correlation r) | 0.85 - 0.95 [1, 2] | N/A | High correlation validates automation for longitudinal % coverage tracking. |
| Key Output Metrics | Plaque % Coverage, Red Plaque %, ΔR value, ΔQ value | Plaque % Coverage, Red Plaque % | Automated provides additional quantitative fluorescence parameters. |
| Susceptibility to Error | Image quality, calibration, threshold setting | Investigator experience, subjective bias, fatigue | Bracket shadows/glare can confound both; standardized lighting is critical. |
| Best Use Case | High-throughput screening, objective longitudinal tracking, multi-center trials. | Gold standard validation, complex cases with heavy staining, algorithm training. | Manual often used as ground truth to train/validate automated systems. |
| Required Expertise | Software operation, basic parameter understanding. | Extensive training in plaque morphology, high intra-rater consistency. |
Sources: [1] Contemporary studies using Inspektor Pro QA2 software. [2] Validation studies against traditional indices (e.g., Modified Quigley-Hein).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for QLF-D Plaque Assessment Protocols
| Item | Function in Protocol | Example/Specification |
|---|---|---|
| QLF-D Imaging Device | Captures quantitative fluorescence images of teeth. | Inspektor Pro with DSLR camera, specific blue-violet LED light (405 nm), and yellow filter. |
| Calibration Target | Ensures consistency of light intensity and color across imaging sessions. | White balance and fluorescence reference standard provided with device. |
| Analysis Software | Processes images, runs detection algorithms, calculates metrics. | Inspektor QA2 v.1.26, ImageJ with custom macros, or Python (OpenCV, scikit-image). |
| Digital Stylus & Tablet | For manual tracing protocol to improve precision and reduce hand fatigue. | Wacom Intuos or similar. |
| Data Management Database | Securely stores raw images, analysis files, and exported data for thesis research. | REDCap, local SQL database, or structured network drive with audit trail. |
| Statistical Software | Analyzes exported plaque metrics for significant differences between study groups. | SPSS, R, or GraphPad Prism. |
Standardized Data Export Protocol
A consistent export format is mandatory for thesis-level analysis. The workflow should feed directly into statistical packages.
Diagram Title: Data Export Pathway for Statistical Analysis
Export Protocol Steps:
- From the analysis software, select "Export Data" or "Save Results."
- Configure export to include the Core Data Fields listed in the diagram.
- Save files in a non-proprietary format (e.g.,
.csv) using a consistent naming convention (e.g.,StudyID_SubjectID_Visit_Date.csv). - Import the CSV files into a central database linked to patient demographic and clinical metadata (e.g., bracket type, oral hygiene regimen).
- Perform quality checks on the imported data for range and logical consistency before statistical analysis.
Integrating a rigorous software workflow for QLF-D image analysis is fundamental for thesis research in orthodontics. While automated detection offers efficiency and objectivity for large-scale longitudinal plaque assessment, manual detection remains a vital tool for validation and complex cases. A standardized protocol encompassing both methods, coupled with a disciplined data export pipeline, ensures the generation of reliable, analyzable data to support robust scientific conclusions on plaque dynamics during orthodontic treatment.
The integration of Quantitative Light-induced Fluorescence-Digital (QLF-D) imaging into clinical orthodontic research necessitates a rigorous study design where the timing of assessments is critically linked to the interpretation of biofilm dynamics and enamel health. This protocol details the standardized timing of QLF-D assessments and their correlation with established clinical parameters to objectively quantify plaque accumulation and demineralization risk during fixed appliance therapy.
Core Experimental Protocol: Longitudinal QLF-D Assessment in Orthodontics
2.1 Objective: To longitudinally monitor changes in plaque fluorescence (ΔR) and enamel demineralization (ΔF) at designated intra-oral sites throughout orthodontic treatment and correlate these with clinical indices.
2.2 Materials & Site Selection:
- QLF-D Device (Inspektor Pro): Calibrated prior to each assessment session.
- Fluorescent Reference Standard: For daily calibration.
- Clinical Examination Kit: Disposable mirrors, probes, compressed air, CPI probe.
- Site Selection: A minimum of 6 index teeth (e.g., 16, 11, 26, 36, 31, 46) are selected. On each tooth, four specific sites around orthodontic brackets are assessed:
- Gingival margin (GM)
- Incisal/Occlusal margin (OM)
- Distal papilla (DP)
- Mesial papilla (MP)
2.3 Assessment Timing Schedule:
| Assessment Timepoint | Clinical Context | Primary QLF-D Metrics | Correlative Clinical Parameters |
|---|---|---|---|
| T0: Baseline | Pre-bonding (or within 1 week post-bonding) | Baseline plaque fluorescence (ΔR0), Baseline enamel fluorescence (ΔF0) | Plaque Index (PI), Gingival Index (GI), Baseline Photography |
| T1: Short-term Follow-up | 4-6 weeks post-bonding/professional prophylaxis | ΔR, ΔF | PI, GI, Bleeding on Probing (BOP) |
| T2: Mid-term Follow-up | 3-4 months (aligned with adjustment visits) | ΔR, ΔF, calculated trends | PI, GI, BOP, White Spot Lesion (WSL) visual assessment |
| T3: Long-term Follow-up | 6-8 months (or pre-debonding) | ΔR, ΔF, area of affected enamel (mm²) | PI, GI, Modified ICDAS for WSLs, Debonding notes |
| T4: Post-Treatment | 1-3 months post-debonding | ΔF (remineralization monitoring) | Post-treatment photography, Final ICDAS |
2.4 Procedural Workflow:
- Patient Preparation: Refrain from oral hygiene for 24 hours prior to plaque assessment. For demineralization-only assessments, conduct professional prophylaxis 1 week prior.
- Clinical Parameter Recording: First, record PI (Silness & Löe) and GI (Löe & Silness) at designated sites.
- QLF-D Image Capture: Dry tooth surface with compressed air for 5 seconds. Capture QLF-D images under standardized conditions (dark room, fixed distance, cheek retractor in place). Ensure the fluorescent reference standard is within the frame.
- Image Analysis: Use proprietary software (QA2 v.1.2+) to analyze regions of interest (ROIs). For plaque, calculate the red fluorescence intensity loss (ΔR, %). For enamel, calculate the fluorescence loss (ΔF, %) and lesion area (mm²).
Correlation Analysis Protocol
3.1 Data Structuring: Organize data in a table format per patient per timepoint.
| Patient ID | Timepoint | Tooth/Site | ΔR (%) | ΔF (%) | Area (mm²) | PI Score | GI Score | BOP (Y/N) |
|---|---|---|---|---|---|---|---|---|
| P-01 | T0 | 16-GM | 12.5 | 0.5 | 0.0 | 1 | 0 | N |
| P-01 | T1 | 16-GM | 45.3 | -2.1 | 0.5 | 2 | 1 | Y |
3.2 Statistical Correlation:
- Primary Analysis: Spearman's rank correlation coefficient (ρ) between ΔR values and corresponding PI scores across all sites and timepoints.
- Secondary Analysis: Linear mixed-effects models to assess the longitudinal relationship between ΔR (predictor) and GI/BOP (outcome), accounting for repeated measures within subjects.
- Tertiary Analysis: Pearson correlation between ΔF at T3/T4 and visual WSL severity scores (ICDAS).
3.3 Expected Quantitative Correlations (Based on Current Literature):
| Correlation Pair | Expected Coefficient Range (ρ/r) | Strength & Significance | Key Reference Insight |
|---|---|---|---|
| ΔR vs. PI (Silness & Löe) | 0.70 - 0.85 | Strong, significant (p<0.001) | QLF-D provides continuous, sensitive data vs. ordinal PI. |
| ΔR (Baseline) vs. GI (6mo) | 0.50 - 0.65 | Moderate, significant (p<0.01) | Early plaque fluorescence predicts later gingival inflammation. |
| ΔF (at Debond) vs. ICDAS Score | 0.75 - 0.90 | Strong, significant (p<0.001) | Validates QLF-D as an objective measure of demineralization severity. |
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in QLF-D Protocol | Example/Specification |
|---|---|---|
| QLF-D System (Inspektor Pro) | Captures auto-fluorescence images of plaque (red) and enamel (green). | Includes camera, LED array (405nm), software, and calibration tool. |
| Fluorescent Reference Standard | Ensures inter- and intra-session imaging consistency and software calibration. | Polyethylene block with embedded fluorescent dye. |
| QA2 Analysis Software | Quantifies ΔR, ΔF, and lesion area from captured images. | Version 1.2 or higher required for advanced plaque analysis. |
| Disposable Cheek Retractors | Provides consistent field of view and prevents soft tissue obstruction. | Single-use, plastic. |
| CPI Probe (WHO) | For standardized clinical assessment of plaque and gingival status. | Ball-end, 0.5mm diameter. |
| Image Archiving Database | Securely stores raw images and linked clinical data for longitudinal analysis. | HIPAA/GDPR-compliant server (e.g., XNAT, local SQL). |
Visualization: Study Workflow & Data Correlation Logic
Diagram Title: Longitudinal QLF-D Study Workflow
Diagram Title: QLF-Correlation Analysis Logic
Optimizing QLF-D Data Quality: Troubleshooting Common Artifacts and Variability in Orthodontic Studies
Accurate plaque quantification using Quantitative Light-induced Fluorescence Digital (QLF-D) technology is critical for assessing oral hygiene efficacy and anti-plaque agent performance in orthodontic research. A primary challenge is the confounding fluorescence or physical obstruction caused by common intraoral factors: saliva, blood, dental calculus, and excess composite resin flash from bracket bonding. This document provides detailed application notes and protocols for managing these interferences within a clinical research protocol, ensuring data integrity for longitudinal orthodontic studies.
Interference Characteristics & Quantitative Impact
The following table summarizes the spectral characteristics and impact of each interfering substance on QLF-D-based plaque assessment.
Table 1: Interference Profile of Common Intraoral Factors in QLF-D Analysis
| Interfering Substance | Primary QLF-D Impact (at 405nm excitation) | Key Fluorescence Signature (Approx. Wavelength) | Effect on Plaque ΔR (Red Fluorescence) Value | Typical Magnitude of ΔR Deviation |
|---|---|---|---|---|
| Saliva (Pooling) | Light scattering, diffusion, and attenuation. Creates uneven illumination. | Weak autofluorescence (broad, 450-550 nm). | Underestimation; reduces contrast. | ΔR decrease: 5-15% in affected zones. |
| Blood (Hemoglobin) | Strong absorption of blue-violet light and emitted fluorescence (Soret band). | Major absorption peaks at ~410-430 nm. | Severe underestimation; can obscure plaque signal entirely. | ΔR decrease: 20-50%+ (highly variable). |
| Calculus | Strong green/white fluorescence, often brighter than plaque. | Intense emission, 520-560 nm (similar to green channel). | Overestimation; misclassified as plaque. | Can mimic ΔR values of 20-40%. |
| Composite Resin Flash | Intense blue/white fluorescence, high reflectivity. | Very strong emission, 430-480 nm (blue channel). | Severe overestimation; saturation of blue channel signal. | ΔR values can exceed 100%, saturating pixels. |
Note: ΔR (ΔRed) is the change in red fluorescence intensity from plaque bacteria relative to the sound tooth surface. Percent deviations are estimates based on recent clinical imaging studies (2023-2024).
Experimental Protocols for Interference Mitigation & Control
Protocol 3.1: Pre-Imaging Oral Cavity Preparation
Objective: Standardize the subject's oral condition to minimize salivary and hemorrhagic interference prior to QLF-D image capture. Materials: Research-grade disposable mirrors, suction tips, 3-in-1 air/water syringe, cotton gauze rolls (2x2 inch), sterile cotton pellets, disclosing solution (if required by protocol). Workflow:
- Instruct subject to gently rinse with water for 10 seconds to clear loose debris.
- Perform a visual screening for gross blood (gingival bleeding) using a disposable mirror and operatory light.
- For saliva control: Isolate the target quadrants using cotton gauze rolls in the vestibule and sublingually. Gently dry tooth surfaces with a 3-second burst of air from the syringe held 1-2 cm perpendicular to the tooth surface. Avoid desiccation.
- For blood control: If minor gingival bleeding is present, apply light pressure with a sterile cotton pellet for 60 seconds. Re-inspect. If bleeding persists, note the site and exclude it from analysis. Postpone imaging for subjects with generalized gingivitis (BOP > 20% sites).
- Proceed to imaging within 30 seconds of drying.
Protocol 3.2: In-Silico Image Correction & Segmentation Workflow
Objective: Process raw QLF-D images to identify and exclude pixels contaminated by calculus or composite resin flash. Software: Custom MATLAB/Python script (e.g., with OpenCV, SciPy) or specialized image analysis suite (e.g., QA2 v.1.30). Methodology:
- Image Alignment & Channel Separation: Align red (R), green (G), and blue (B) fluorescence channels from the raw QLF-D image.
- Composite Resin Flash Detection: Identify pixels in the Blue (B) channel where intensity exceeds a calibrated threshold (e.g., >95% of dynamic range). These high-intensity, sharply bordered regions are classified as "composite flash."
- Calculus Detection: Calculate a G/R ratio for each pixel. Pixels with a G/R ratio exceeding a set threshold (e.g., >1.5) and absolute G intensity above plaque background are flagged as "calculus."
- Mask Application & Data Exclusion: Generate binary masks for "composite flash" and "calculus." Apply these masks to the ΔR analysis map. Exclude all masked pixels from the final plaque quantification.
- Output: Report total plaque area (ΔR > 5%) and mean ΔR value from valid, uncontaminated pixels only.
Visualization of Experimental and Analytical Workflows
Diagram 1: QLF-D Imaging and Analysis Workflow for Interference Management
Diagram 2: Light Interaction and Interference with QLF-D Signals
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Research Reagent Solutions for Controlled QLF-D Studies
| Item Name | Specification / Brand Example | Primary Function in Protocol | Critical Notes for Research |
|---|---|---|---|
| QLF-D Clinical Imaging System | Inspektor Pro with QA2 Software | Gold-standard device for capturing quantitative fluorescence images of plaque (ΔR). | Ensure consistent calibration with reference standard before each imaging session. |
| Disclosing Solution (if used) | Two-Tone (e.g., 1.5% Bismarck Brown, 4.5% Fast Green) | Visually distinguishes mature (blue) vs. new (pink) plaque for validation. | Use sparingly. May temporarily alter fluorescence; allow 24h washout before QLF-D. |
| Synthetic Saliva / Wetting Agent | Xerostomia relief solutions (e.g., Biotène formulations) | Simulate standardized salivary film for in-vitro or ex-vivo model validation. | Useful for testing the limits of saliva interference in a controlled setting. |
| Hemoglobin Standard Solution | Lyophilized human hemoglobin, reconstituted. | Create calibrated absorption phantoms to quantify blood interference thresholds. | For in-vitro model development only. |
| Reference Calculus Samples | Extracted human teeth with supra-gingival calculus. | Provide positive controls for calibrating calculus detection algorithms. | Scan with micro-CT to correlate fluorescence with mineral density. |
| Orthodontic Composite Resin | Transbond XT or equivalent. | Create standardized "flash" samples on enamel or acrylic slabs for thresholding. | Cure to manufacturer specifications. Different brands have varying fluorescence. |
| Image Analysis Software | MATLAB with Image Processing Toolbox, Python (OpenCV, SciPy), or QA2. | Implement custom pixel classification, masking, and quantification algorithms. | Essential for executing Protocol 3.2. Requires programming expertise. |
1. Introduction
This application note outlines protocols to mitigate three critical sources of technical variability in Quantitative Light-induced Fluorescence (QLF) research, specifically within a clinical thesis focused on longitudinal plaque assessment during orthodontic treatment. Reliable quantification of red and green fluorescence, crucial for evaluating cariogenic activity and therapeutic efficacy, is highly sensitive to operator technique, device performance stability, and image acquisition quality. Failure to control these variables introduces noise that can obscure true biological signals, compromising data integrity in multi-center trials or longitudinal studies.
2. Research Reagent Solutions & Essential Materials
Table 1: Key Research Reagent Solutions for QLF-D Plaque Assessment
| Item | Function in QLF-D Protocol |
|---|---|
| QLF-D Biluminator 2+ (Inspektor Research) | Dual blue-violet (405 nm) and white light LED source for quantitative fluorescence and visual imaging. Primary research device. |
| QLF-D Calibration Standard (BaSO₄/MgO puck) | A stable, non-fluorescent reference standard for daily white balance calibration to correct for illumination intensity drift. |
| Fluorescent Reference Puck (e.g., RB 220) | A stable, homogenous fluorescent target for periodic validation of fluorescence quantification linearity and system response. |
| Automated Alignment Jig (e.g., C4) | A patient positioning system with chin and forehead rest to standardize camera-to-subject distance (≈40 mm) and angle (90°). |
| Image Acquisition Software (e.g., QA2 v2.0+) | Software controlling capture parameters (exposure, gain) and enabling live focus assessment tools (e.g., Focus Index). |
| Demineralized Enamel Phantoms | Laboratory-grade samples with controlled lesion severity for periodic validation of the ΔQ (fluorescence loss) calculation algorithm. |
3. Protocols for Mitigating Technical Variability
3.1 Protocol for Operator Training and Consistency Certification Objective: To achieve high inter- and intra-operator reliability in image capture. Methodology:
- Theoretical Training: Operators complete modules on QLF-D principles, plaque indices (e.g., modified Quigley-Hein), and orthodontic appliance-specific challenges.
- Practical Calibration:
- Using the alignment jig, the operator captures 10 repeated QLF images of a typodont with fixed orthodontic brackets and simulated plaque.
- Images are analyzed for consistent coverage (≥95% of target tooth in frame), focus, and absence of saliva pooling or glare.
- Certification Metric: An operator is certified when their coefficient of variation (CV) for the calculated ΔQ value of a reference plaque region across 10 images is <5%.
3.2 Protocol for Managing Device Calibration Drift Objective: To ensure longitudinal stability of fluorescence intensity measurements. Methodology:
- Daily White Balance Calibration: Before each imaging session, capture an image of the BaSO₄/MgO calibration puck under white light mode. Software normalizes subsequent images based on this reference.
- Weekly Fluorescence Validation:
- Capture an image of the stable fluorescent reference puck (RB 220).
- Extract the mean red (F630) and green (F525) fluorescence values from a standard Region of Interest (ROI).
- Record values in a control chart (see Table 2).
- Corrective Action: If values drift beyond ±10% from the established baseline, initiate device service and suspend research imaging.
Table 2: Example Control Chart for Fluorescence Validation
| Date | F525 Mean (a.u.) | % Deviation from Baseline | F630 Mean (a.u.) | % Deviation from Baseline | Action |
|---|---|---|---|---|---|
| Baseline | 150.2 | 0.0% | 89.5 | 0.0% | -- |
| 2023-10-10 | 149.8 | -0.3% | 90.1 | +0.7% | None |
| 2023-10-17 | 138.5 | -7.8% | 81.0 | -9.5% | Re-calibrate, then re-test. |
| 2023-10-18 (Post-Cal) | 150.5 | +0.2% | 89.8 | +0.3% | Device returned to service. |
3.3 Protocol for Standardizing Image Focus Objective: To eliminate focus variability as a source of error in fluorescence quantification. Methodology:
- Jig-Based Positioning: Mandatory use of the automated alignment jig for all subject imaging.
- Live Focus Assessment: Utilize the software's real-time Focus Index (FI) or edge sharpness tool. The operator must adjust fine-focus until the FI target (e.g., >85) is met.
- Post-Capture Quality Check: Implement a standard operating procedure (SOP) where every image is reviewed against criteria before the subject is dismissed.
- Accept: All tooth edges in the target ROI are sharply defined. FI > 85.
- Reject: Any blur in the ROI. Presence of saliva bubbles or specular reflection obstructing the surface. Immediate re-capture required.
4. Integrated Workflow and Data Integrity Pathway
QLF-D Clinical Imaging Integrity Workflow
5. Experimental Protocol for Validating the Full Control System Objective: To empirically demonstrate that the implemented protocols reduce technical variability. Design: A controlled, cross-over study.
- Subjects: 5 typodont models with bonded brackets and standardized, fluorescent simulated plaque.
- Phase 1 (Uncontrolled): 3 untrained operators capture 5 images each per typodont without using jigs, focus protocols, or calibration checks.
- Phase 2 (Controlled): The same operators complete certification, then capture 5 images each per typodont using the full protocol (jig, focus index, post-calibration).
- Analysis: Calculate the Intra-class Correlation Coefficient (ICC) and CV for ΔQ measurements within and between operators for each phase.
Table 3: Expected Outcome of Validation Experiment
| Metric | Phase 1 (Uncontrolled) | Phase 2 (Controlled) | Interpretation |
|---|---|---|---|
| Inter-operator ICC | Poor (<0.50) | Excellent (>0.90) | Protocol ensures different operators get the same result. |
| Intra-operator CV | High (>15%) | Low (<5%) | Protocol ensures the same operator is consistent over time. |
| Overall ΔQ Range | Wide (e.g., 50-200) | Narrow (e.g., 120-140) | Total technical noise is drastically reduced. |
Conclusion The rigorous application of these standardized protocols for operator certification, systematic calibration monitoring, and focus control is non-negotiable for producing high-fidelity, reproducible QLF-D data in orthodontic plaque research. This framework directly supports the integrity of a clinical thesis by isolating biological variability from technical artifact, thereby strengthening the validity of conclusions regarding plaque dynamics and treatment effects.
Within the research framework for establishing a standardized Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocol for plaque assessment during orthodontic treatment, the physical and material properties of different bracket systems present significant methodological challenges. The bracket type directly influences plaque adhesion, biofilm architecture, and fluorescence signals, potentially confounding longitudinal plaque quantification. This note details the application challenges and necessary experimental controls when integrating QLF-D assessment across ceramic, metal, and self-ligating bracket systems.
The following table summarizes key challenges and optical interference data associated with each bracket type, derived from recent studies on fluorescence imaging and biofilm retention.
Table 1: Comparative Challenges of Bracket Types in QLF-D Plaque Assessment
| Bracket Type | Primary Challenge for QLF-D | Average Plaque Retention Index | Fluorescence Interference | Notes on Biofilm Architecture |
|---|---|---|---|---|
| Ceramic | High autofluorescence (green-blue spectrum) mimicking bacterial porphyrins. | 1.8 (Est.) | High (Can mask red plaque fluorescence) | Smooth surface promotes sheet-like biofilm; translucent brackets allow sub-surface scattering. |
| Metal (Stainless Steel) | Specular reflection; opaque nature casts shadows, altering light homogeneity. | 2.1 (Est.) | Low (Minimal autofluorescence) | Surface imperfections (etching) promote complex, stratified biofilm colonies. |
| Self-Ligating | Design complexity (clip/doors) creates niche areas inaccessible to standard imaging angles. | Passive: 1.9Active: 2.3 | Varies (Depends on constituent material) | Gate mechanism creates a protected, anaerobic environment for biofilm maturation. |
Note: Plaque Retention Index estimates are based on a scale of 0-3 (0=low, 3=high), synthesized from recent *in situ studies. Fluorescence Interference refers to the bracket's intrinsic signal noise against QLF-D's detection of bacterial porphyrins.*
Experimental Protocols for Controlled QLF-D Assessment
Protocol 1: Baseline Autofluorescence Characterization of Bracket Materials Objective: To map and quantify the intrinsic fluorescence signature of uncontaminated bracket systems across the QLF-D emission spectrum. Materials: Unused ceramic, metal, and self-ligating brackets (n=10 per type), QLF-D imaging system (Inspektor Pro or equivalent), spectral calibration tile, dark enclosure. Methodology:
- Calibration: Perform white balance and spectral calibration of the QLF-D system using the provided tile in a completely dark environment.
- Imaging: Mount each bracket on a non-fluorescent black background. Acquire QLF-D images using standard blue-violet light excitation (405 nm).
- Analysis: Using proprietary software (QA2), analyze Regions of Interest (ROI) on the bracket body, wing, and ligation mechanism. Record fluorescence intensity (ΔF, ΔR) values across the red (porphyrin) and green spectral channels.
- Documentation: Create a reference library of autofluorescence profiles for each bracket type/model to be subtracted from subsequent in vivo images.
Protocol 2: In Situ Plaque Accumulation and QLF-D Analysis with Bracket-Specific ROIs Objective: To standardize plaque quantification on teeth while accounting for bracket-induced optical artifacts. Materials: Orthodontic patients with informed consent, characterized bracket systems, QLF-D system, plaque disclosure solution (e.g., erythrosine). Methodology:
- Patient Preparation: Refrain from oral hygiene for 24 hours prior to imaging.
- Bracket Mapping: Prior to bonding, photograph and digitally map the exact position and type of each bracket in the arch.
- QLF-D Imaging Session: a. Acquire pre-disclosure QLF-D images, ensuring the camera is positioned perpendicular to the tooth surface to minimize reflection. b. Apply disclosing agent, rinse gently. c. Acquire post-disclosure QLF-D images.
- Image Processing & Analysis: a. Import images. Using the bracket map, apply material-specific autofluorescence correction filters from Protocol 1. b. Define two sets of ROIs: Standard Enamel ROI (1mm away from bracket perimeter) and Perimeter ROI (0.5mm zone adjacent to bracket base). c. Quantify plaque severity (ΔR) and area (%) for each ROI. Note shadowed areas from metal brackets and clip areas in self-ligating systems as "masked zones" excluded from primary analysis.
Visualizations
Diagram 1: QLF-D Assessment Workflow with Bracket Correction
Diagram 2: Bracket-Specific Challenge Pathways
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Controlled QLF-D Orthodontic Research
| Item | Function in Protocol | Specification/Note |
|---|---|---|
| QLF-D Clinical System | Primary imaging device for plaque fluorescence quantification. | Must have capacity for multi-wavelength excitation (e.g., 405 nm) and validated software (QA2) for ΔR calculation. |
| Spectral Calibration Tile | Ensures consistency and accuracy of fluorescence measurements across imaging sessions. | Use manufacturer-provided tile for daily calibration under dark conditions. |
| Non-Fluorescent Mounting Medium | Holds brackets for in vitro characterization without adding fluorescent noise. | e.g., Black dental wax or non-fluorescent epoxy. |
| Plaque Disclosing Solution (Erythrosine) | Provides visual validation of QLF-D plaque detection areas post-imaging. | Use at low concentration (e.g., 0.5%) to avoid quenching natural fluorescence. |
| Bracket Reference Library Kit | Physical set of all bracket types/models used in the clinical study. | Used for pre-study autofluorescence mapping (Protocol 1). |
| ROI Mapping Software Module | Allows precise definition of bracket-adjacent and standard enamel areas for analysis. | Should integrate with QLF-D software; capable of handling digital bracket maps. |
| Matte-Finish Positioning Aid | Reduces ambient light and standardizes camera-tooth distance and angle. | Custom-fabricated mouth prop with matte black surface to prevent reflection. |
1. Introduction within the QLF-D Clinical Protocol Thesis Context
This document details the application notes and protocols for software parameter tuning, specifically the adjustment of fluorescence threshold settings, to distinguish between different stages of dental plaque maturation. This protocol is an integral component of a broader thesis developing a standardized Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocol for longitudinal plaque assessment in orthodontic patients. Accurate software-based staging is critical for quantifying biofilm dynamics in response to oral hygiene interventions, antimicrobial therapies, or biomaterial interactions during fixed-appliance therapy.
2. Core Principles: Plaque Maturation and QLF-D Signal
Plaque maturation progresses from early, thin, reversible colonies to mature, complex biofilms. QLF-D (Inspektor Pro, Amsterdam) exploits the loss of red fluorescence (due to bacterial porphyrins) and the increase in green fluorescence as plaque accumulates and matures. The software's analysis algorithm classifies pixels based on the ratio of red-to-green fluorescence ((\Delta R)) and the total loss of fluorescence ((\Delta F)).
- Early/Immature Plaque: Higher red fluorescence, lower (\Delta F).
- Mature Plaque: Lower red fluorescence, higher green fluorescence, higher (\Delta F).
The software's built-in "Plaque Mode" uses default thresholds. For research, especially in orthodontics where plaque accumulation patterns are altered by brackets and wires, tuning these thresholds is essential to match the visual clinical assessment (e.g., modified Quigley-Hein index) and to sensitively detect subtle changes between study timepoints.
3. Quantitative Data Summary: Suggested Threshold Ranges
Based on a synthesis of current literature and calibration studies, the following threshold ranges are proposed for initial tuning. Final values must be validated per institutional protocol.
Table 1: Suggested QLF-D Software Threshold Parameters for Plaque Staging
| Plaque Stage (QLF-D Classification) | (\Delta R) Threshold (Red Fluorescence Loss) | (\Delta F) Threshold (Total Fluorescence Loss) | Visual/Clinical Correlation |
|---|---|---|---|
| Sound Enamel / Background | (\Delta R) < 0.10 | (\Delta F) > -5% | No visible plaque. |
| Early/Immature Plaque | 0.10 ≤ (\Delta R) < 0.35 | -30% ≤ (\Delta F) ≤ -5% | Thin, patchy plaque, disclosing solution: light pink. |
| Mature/Biofilm Plaque | (\Delta R) ≥ 0.35 | (\Delta F) < -30% | Thick, structured biofilm, disclosing solution: dark blue/purple. |
Note: (\Delta R) is a unitless ratio. (\Delta F) is a percentage change relative to the sound enamel reference fluorescence.
4. Experimental Protocol: Calibration and Validation of Thresholds
Protocol 4.1: In-Vitro Threshold Calibration Using Standardized Panels Objective: To establish a baseline correlation between fluorescence metrics and known plaque thickness/maturity. Materials:
- QLF-D system with research software (QA2 v1.20 or later).
- Hydroxyapatite discs with cultured Streptococcus mutans and Porphyromonas gingivalis biofilms of known age (24h, 48h, 72h, 7d).
- Microplate reader for bacterial cell count (OD600) and confocal laser scanning microscopy (CLSM) for biofilm thickness validation. Method:
- Image each biofilm disc (n=10 per timepoint) under standardized QLF-D conditions (distance, angle, exposure).
- Analyze using default software settings. Record mean (\Delta R) and (\Delta F) for the region of interest (ROI).
- Correlate (\Delta R) and (\Delta F) values with biofilm age, cell count, and CLSM-measured thickness/bivolume.
- Use receiver operating characteristic (ROC) analysis to determine the (\Delta R) and (\Delta F) values that best discriminate between 24h (immature) and 7d (mature) biofilms. These values become your initial tuned thresholds.
Protocol 4.2: In-Vivo Validation Against Clinical Indices in Orthodontic Patients Objective: To validate tuned thresholds against a clinical plaque index in the target population. Materials:
- QLF-D system with tunable software.
- Orthodontic patients (ethics-approved).
- Disclosing solution (e.g., 2-tone). Method:
- Prior to a scheduled adjustment, photograph the labial surfaces of teeth 11-16, 31-36 (or full arch) using QLF-D.
- Have a calibrated examiner immediately score the same surfaces using the modified Quigley-Hein (Turesky) plaque index (0-5).
- In the software, apply the tuned thresholds from Protocol 4.1. Calculate the percentage of plaque-covered area per tooth classified as "Mature."
- Perform statistical correlation (Spearman's rank) between the software-derived "% Mature Plaque Area" and the clinical index score.
- Iteratively adjust thresholds within the ranges from Table 1 to maximize the correlation coefficient. The final, validated thresholds are documented as the lab's standard operating procedure (SOP) for the study.
5. Visualization of Analysis Workflow
Diagram Title: QLF-D Image Analysis Workflow for Plaque Staging
6. The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials for QLF-D Plaque Staging Research
| Item | Function in Protocol | Example/Specification |
|---|---|---|
| QLF-D Research Device | Captures quantitative fluorescence images. Must allow raw data export and parameter adjustment. | Inspektor Pro with QA2 Research Software. |
| Calibration Standards | Ensures day-to-day instrument reproducibility and cross-site comparability. | Ceramic fluorescence reference slab (supplied). |
| 2-Tone Disclosing Solution | Provides visual clinical reference for plaque maturity (pink=early, blue=mature). | Contains erythrosin (pink) & fast green (blue). |
| Hydroxyapatite Discs | In-vitro substrate for growing standardized, reproducible biofilms for threshold calibration. | Sintered, saliva-coated, diameter 5-10mm. |
| Confocal Laser Scanning Microscope (CLSM) | Gold-standard for validating biofilm 3D structure, thickness, and biovolume in calibration studies. | With LIVE/DEAD BacLight viability staining. |
| Statistical Analysis Software | For ROC analysis, correlation studies, and longitudinal data modeling. | R, Python (SciPy), or GraphPad Prism. |
| Orthodontic Study Model | For in-vitro simulation of bracket/wire environment during method development. | Typodont with bonded brackets and archwires. |
This document details the rigorous data integrity framework implemented within a thesis investigating Quantitative Light-induced Fluorescence-Digital (QLF-D) for dental plaque assessment during fixed orthodontic treatment. The protocols ensure the reliability, objectivity, and traceability of clinical plaque quantification data, which is critical for evaluating anti-plaque agents or oral hygiene interventions in orthodontic research.
Blinding (Masking) Protocols
Application Notes
Blinding is essential to minimize performance and detection bias. In a typical QLF-D orthodontic study, multiple parties require masking to prevent conscious or subconscious influence on results.
Detailed Protocol: Triple-Blind Study Design
Objective: To blind the participant, clinical examiner/image capturer, and data analyst independently.
Materials: QLF-D imaging system (e.g., QLF-D Biluminator 2+), patient-specific random code generator, secure database, opaque image processing software overlays.
Workflow:
- Participant Blinding: Participants are not informed which treatment arm (e.g., test mouthwash vs. placebo control) they are assigned to. All investigational products are identical in appearance, taste, and packaging.
- Examiner/Image Capturer Blinding: The clinician performing plaque disclosure and QLF-D image capture is kept unaware of the participant's group allocation. A separate study coordinator handles product distribution.
- Analyst Blinding: All QLF-D images are de-identified using a randomized alphanumeric code (e.g., P-045-T2). The analyst uses software (e.g., QA2 v2.0.0.27 software) where the code is displayed, but no links to participant ID, visit, or treatment group are visible during quantification.
Integrity Check: Unblinding occurs only after database lock and completion of the primary statistical analysis.
Inter- and Intra-Examiner Reliability Assessments
Application Notes
QLF-D analysis involves manual selection of regions of interest (ROIs) on teeth, requiring assessment of consistency within (intra-) and between (inter-) analysts to ensure data precision.
Detailed Protocol: Reliability Calibration for QLF-D Plaque Analysis
Objective: To establish and document a minimum threshold of agreement (≥0.90 Intraclass Correlation Coefficient) for plaque fluorescence loss (ΔF) and area measurements.
Materials: Calibration set of 30 anonymized QLF-D images (representing various plaque levels around brackets/gingiva), QLF-D analysis software, statistical software (e.g., SPSS, R).
Pre-Training: All analysts undergo standardized training on ROI placement for orthodontic patients (e.g., defining borders at the bracket gingival margin, excluding gingival tissue).
Procedure for Intra-Examiner Reliability:
- Analyst A quantifies the calibration image set at Time 1 (T1).
- After a minimum washout period of 14 days, Analyst A re-quantifies the same image set in a randomized order at Time 2 (T2).
- ΔF and plaque area values from T1 and T2 are compared using Intraclass Correlation Coefficient (ICC) with a two-way mixed-effects, absolute agreement model.
Procedure for Inter-Examiner Reliability:
- Analysts A, B, and C each quantify the same calibration image set independently.
- Their results for ΔF and plaque area are compared using ICC with a two-way random-effects, absolute agreement model.
Acceptance Criteria: ICC ≥ 0.90 for both ΔF and area for each examiner (intra) and between the mean scores of all examiners (inter). Analysts failing this threshold undergo re-training and re-assessment.
Table 1: Example Reliability Assessment Results
| Examiner | Metric | ICC (95% CI) vs. Gold Standard | Agreement Level |
|---|---|---|---|
| Intra-Examiner A | ΔF | 0.96 (0.92 - 0.98) | Excellent |
| Intra-Examiner A | Area | 0.93 (0.86 - 0.97) | Excellent |
| Inter-Examiner (A,B,C) | ΔF | 0.91 (0.84 - 0.95) | Excellent |
| Inter-Examiner (A,B,C) | Area | 0.89 (0.81 - 0.94) | Good |
Audit Trail Implementation
Application Notes
A comprehensive audit trail provides a secure, time-stamped record of all data-related activities, from image acquisition to statistical analysis, ensuring reconstruction of the study events.
Detailed Protocol: End-to-End Data Traceability
Objective: To maintain a complete, immutable record of all data creation, modification, and access.
System Components: Electronic Case Report Form (eCRF), secure server with access logs, version-controlled database, standardized file naming convention, electronic lab notebook (ELN).
Key Protocol Steps:
- Data Origin: QLF-D images are automatically saved with metadata: Participant Random ID, Visit Date/Time, Device Serial Number, Camera Settings.
- Data Processing: Any image adjustment (e.g., normalization) is performed on a copy. The original file is preserved. The analysis software log records the analyst ID, date/time of analysis, and software version for each saved ROI data file.
- Data Entry & Changes: All data entered into the eCRF or master database is logged. Any change captures the previous value, new value, reason for change, and user ID.
- Analysis Trail: Statistical scripts (e.g., R Markdown, Jupyter Notebooks) are used, providing a reproducible record of all data manipulations and analytical steps.
Table 2: Critical Elements of the QLF-D Study Audit Trail
| Process Stage | Recorded Element | Purpose |
|---|---|---|
| Image Capture | Timestamp, Operator ID, Device ID | Links raw data to operator and conditions. |
| Data Transfer | Checksum verification log | Ensures file integrity during transfer. |
| Analysis | Analyst ID, Software Version, Timestamp of result file | Attributes derived data to a specific action. |
| Database Change | Old value, New value, Editor ID, Reason | Provides full history of data evolution. |
| Access | Server/File access logs | Detects unauthorized access attempts. |
The Scientist's Toolkit: QLF-D Plaque Assessment Research Reagents & Materials
Table 3: Essential Research Reagent Solutions for QLF-D Orthodontics Studies
| Item | Function in QLF-D Protocol |
|---|---|
| Fluorescein-based Plaque Disclosing Solution (e.g., FD&C Red No. 3) | Selectively stains mature bacterial plaque, inducing fluorescence quenching detectable by QLF-D. Essential for standardizing plaque visibility. |
| Calibration Standards (Ceramic or Resin) | Used for daily or weekly calibration of the QLF-D device to ensure consistent light intensity and camera sensitivity across all study visits. |
| Anti-fogging Solution for Intraoral Camera Lens | Prevents lens fogging during intraoral imaging, ensuring consistent image clarity and quality. |
| Sterile Water & Air Syringe Triplet | For drying the tooth surface prior to QLF-D image capture, as excess saliva can interfere with plaque fluorescence measurements. |
| QA2 or Proprietary QLF-D Analysis Software | Enables standardized quantification of plaque fluorescence loss (ΔF, %), area, and thickness (ΔR) within user-defined ROIs around orthodontic brackets. |
Visualized Protocols and Workflows
QLF-D Triple-Blinding Workflow
Reliability Calibration & Certification Loop
Audit Trail: Data Chain of Custody
Validating QLF-D Outcomes: Correlation with Microbiological and Biochemical Benchmarks
Within the thesis investigating the QLF-D (Quantitative Light-induced Fluorescence-Digital) clinical protocol for plaque assessment during orthodontic research, establishing robust correlations between clinical imaging metrics and underlying microbiological and biochemical parameters is paramount. This document details the application and protocols for conducting correlative studies that link the ΔR value (the calculated red fluorescence loss from QLF-D, indicative of plaque activity and maturity) to direct measures of biofilm microbiology (via Colony Forming Unit counts and quantitative PCR) and plaque pH. The integration of these data streams validates QLF-D as a non-invasive, quantitative research tool for monitoring biofilm dynamics in response to orthodontic appliances, antimicrobial agents, or oral hygiene interventions.
Experimental Protocols
Integrated Clinical Sample Collection & QLF-D Imaging Protocol
Objective: To standardize the collection of plaque biofilm samples from specific orthodontic sites (e.g., bracket periphery, gingival margin) concurrent with QLF-D image acquisition.
- Site Selection: Identify and mark ≥3 representative test sites per subject (e.g., premolar brackets). Include a control site (sound enamel, if available).
- Pre-imaging: Subjects refrain from oral hygiene for 24h. Isolate site with cotton rolls, dry gently with air syringe for 3s.
- QLF-D Imaging: Acquire QLF-D image using standardized settings (e.g., aperture f/4.0, ISO 400, distance 30mm). Ensure the full test site is within the frame. Save image in proprietary format for ΔR analysis.
- Plaque Sampling: Immediately after imaging, using a sterile micro-curette (e.g., Gracey 1/2), harvest plaque from the exact same imaged site. Split the sample into three aliquots in pre-labeled, sterile microcentrifuge tubes for: a) CFU, b) qPCR, c) pH. Process within 30 minutes.
Microbiological Processing: CFU Enumeration
Objective: To quantify total viable aerobic and anaerobic cultivable bacteria from the sampled plaque.
- Homogenization: Vortex the plaque sample in 1 mL of Reduced Transport Fluid (RTF) for 60s. Perform serial 10-fold dilutions (10⁻¹ to 10⁻⁶) in RTF.
- Plating: Plate 100 µL of appropriate dilutions (e.g., 10⁻³, 10⁻⁴, 10⁻⁵) onto Blood Agar (for total aerobes/anaerobes) and Mittal Salivarius Agar (for streptococci). Perform in triplicate.
- Incubation: Incubate Blood Agar plates anaerobically (80% N₂, 10% H₂, 10% CO₂) at 37°C for 7 days. Incubate MSB plates aerobically at 37°C for 48h.
- Counting: Count colonies with distinct morphologies. Calculate CFU/mL of original sample, then normalize to CFU/µg of plaque protein (using a Bradford assay) or per sample site.
Molecular Microbiology: Quantitative PCR (qPCR)
Objective: To quantify total bacterial load and specific cariogenic pathogens (Streptococcus mutans, Lactobacillus spp.).
- DNA Extraction: Use a commercial microbial DNA extraction kit. Include mechanical lysis step (bead-beating) for 90s. Elute in 50 µL TE buffer. Verify purity (A260/A280 ~1.8).
- Primer/Probe Sets: Use validated, published primer-probe sets.
- Total Bacteria: Universal 16S rRNA gene (e.g., 338F/518R).
- S. mutans: gtfB gene-specific.
- Lactobacillus spp.: 16S rRNA group-specific.
- qPCR Reaction: Use a master mix with dUTP and UNG to prevent amplicon carryover. Set up 20 µL reactions in triplicate per sample. Use a standard curve (10²–10⁸ copies of cloned target gene) on every plate.
- Analysis: Express results as log10 gene copy number per µL of DNA eluate, normalized to sample collection volume or protein content.
Plaque pH Measurement
Objective: To measure the resting pH and glycolytic pH drop of the sampled plaque.
- Resting pH: Place the plaque aliquot on a flat-bottom micro pH electrode. Record stable pH value.
- pH Drop Test (Stephen Curve): Add 10 µL of 10% glucose solution to the plaque sample. Record pH every minute for 5-10 minutes. Calculate minimum pH (pH_min) and area under the curve (AUC) for pH < 6.0.
Table 1: Example Correlation Matrix (Hypothetical Data from Orthodontic Cohort, n=50 sites)
| Metric | Mean (±SD) | Correlation with ΔR (Pearson's r) | p-value |
|---|---|---|---|
| QLF-D ΔR Value | 15.3 (±8.7) | 1.00 | N/A |
| Total CFU (log10) | 6.2 (±0.9) / µg protein | 0.78 | <0.001 |
| S. mutans qPCR | 5.1 (±1.2) log copies | 0.82 | <0.001 |
| Lactobacillus qPCR | 4.5 (±1.0) log copies | 0.71 | <0.001 |
| Resting pH | 6.8 (±0.4) | -0.65 | <0.001 |
| Minimum pH | 5.2 (±0.6) | 0.70 | <0.001 |
Table 2: Key Research Reagent Solutions & Materials
| Item / Reagent | Function / Application |
|---|---|
| QLF-D Biluminator 2 | Camera and light source system for standardized plaque fluorescence imaging (ΔR calculation). |
| Reduced Transport Fluid (RTF) | Anaerobic transport medium for preserving viability of plaque bacteria during sample processing. |
| Blood Agar & MSB Agar | Non-selective and selective culture media for total cultivable bacteria and streptococci, respectively. |
| Microbial DNA Extraction Kit | For standardized, high-yield genomic DNA isolation from heterogeneous plaque biofilms. |
| TaqMan Universal PCR MM | Optimized master mix for probe-based qPCR, ensuring high sensitivity and specificity for target genes. |
| Micro pH Electrode (Flat tip) | Enables direct pH measurement of small, viscous plaque samples with minimal sample volume. |
| Sterile Micro-curettes | For precise, site-specific plaque sampling from defined orthodontic surfaces. |
Mandatory Visualizations
Diagram Title: Integrated Workflow for Correlating QLF-D ΔR with Biofilm Analyses
Diagram Title: Logical Links Between Biofilm Activity, pH, and QLF-D ΔR
This application note provides detailed protocols for the comparative benchmarking of digital plaque assessment tools within the framework of a broader thesis on Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocol standardization for orthodontic research. The objective is to establish standardized methodologies for evaluating the performance of emerging technologies—specifically, intraoral scanners (IOS) with integrated plaque detection algorithms and dedicated fluorescence cameras—against the validated reference of QLF-D. This comparison is critical for researchers validating new digital endpoints in plaque quantification and for drug development professionals assessing anti-plaque efficacy in clinical trials.
Table 1: Core Technical Specifications & Measurement Principles
| Parameter | QLF-D (Reference Standard) | Fluorescence Cameras (e.g., VistaCam) | Intraoral Scanners with Plaque Detection (e.g., TRIOS, iTero) |
|---|---|---|---|
| Primary Technology | Blue-violet light (405 nm) fluorescence | Blue light (450 nm) fluorescence | White LED structured light / confocal microscopy + AI algorithm |
| Measured Signal | Loss of green-red fluorescence from plaque bacterial metabolites (porphyrins) | Red fluorescence from bacterial porphyrins | 3D topography + calculated plaque index based on texture/color |
| Primary Output | ΔR (% reduction in fluorescence), plaque area (%) | Fluorescence score (0-3) or area (%) | Plaque Index (PI) score (e.g., 0-5), 3D plaque map |
| Quantification Basis | Pixel-based fluorescence intensity thresholding | Visual or automated scoring of red fluorescence | Surface texture/color deviation from "clean enamel" baseline |
| Validation Status | Extensive peer-reviewed validation for plaque quantification | FDA cleared; moderate clinical validation | Emerging; algorithm-specific, often proprietary validation |
Table 2: Reported Performance Metrics from Recent Studies (2020-2024)
| Benchmarking Study (Example) | Correlation to QLF-D (ΔR) | Correlation to TQPI (Traditional Quigley-Hein) | Key Advantage | Noted Limitation |
|---|---|---|---|---|
| IOS (AI-based) vs. QLF-D | r = 0.72 - 0.89 (strong) | r = 0.78 - 0.92 (strong) | Provides 3D volumetric plaque data; integrates with digital workflow | Algorithm "black box"; sensitive to saliva, staining, restoration artifacts |
| Fluorescence Camera vs. QLF-D | r = 0.85 - 0.95 (very strong) | r = 0.80 - 0.88 (strong) | Real-time visual feedback; simple operation | 2D imaging only; less sensitive to early/biofilm plaque than QLF-D |
| IOS vs. Fluorescence Camera | r = 0.65 - 0.79 (moderate-strong) | r = 0.70 - 0.85 (strong) | No disclosing agent needed; captures full arch | Lower contrast for plaque vs. gum/restorations |
Experimental Protocols for Comparative Benchmarking
Protocol 3.1: In-Vivo Cross-Sectional Comparison in Orthodontic Patients
Objective: To compare the plaque quantification outputs of IOS, fluorescence camera, and QLF-D on bonded orthodontic appliances in a single visit.
Materials (Research Reagent Solutions):
- Subject Cohort: Orthodontic patients (fixed appliances >6 months), n≥30.
- Digital Tools: QLF-D system (Inspektor Pro), fluorescence camera (e.g., Dürr VistaCam), intraoral scanner with plaque software (e.g., 3Shape TRIOS with Plaque Detection).
- Reference Standard: TQPI after disclosing (2-tone solution).
- Software: ImageJ with dedicated plugins (QLF), manufacturer-specific analysis suites.
- Calibration Standards: Fluorescence reflectance standard (Spectralon), intraoral scanner calibration block.
Procedure:
- Ethics & Preparation: Obtain IRB approval and informed consent. Participants abstain from oral hygiene for 24 hours prior.
- Image Acquisition Sequence (Maxillary Arch): a. Step 1: IOS Scan. Perform a full-arch digital scan per manufacturer's protocol. Export the 3D model and plaque index map. b. Step 2: Fluorescence Camera Image. Capture standardized images of buccal surfaces of teeth #16 to #26 using a cheek retractor and positioning stand. c. Step 3: QLF-D Image. Capture identical view as Step 2 using QLF-D in a darkened room. Ensure consistent camera-to-tooth distance. d. Step 4: TQPI Reference. Apply disclosing solution, rinse, and capture standard photographic images for manual TQPI scoring by two blinded examiners.
- Region of Interest (ROI) Alignment: Using image analysis software, digitally align ROIs (e.g., individual tooth surfaces) across all three digital image sets (Fluorescence, QLF-D, clinical photo).
- Data Extraction: For each aligned ROI, extract: IOS Plaque Index, Fluorescence Score, QLF-D ΔR and Area%, and mean TQPI score.
- Statistical Analysis: Perform Pearson/Spearman correlation analysis between each digital metric and TQPI, and between the digital metrics themselves. Apply Bland-Altman analysis to assess limits of agreement.
Protocol 3.2: Longitudinal Monitoring of Anti-Plaque Intervention
Objective: To assess sensitivity to change of each tool in a longitudinal study evaluating a chemical anti-plaque agent.
Materials: As per Protocol 3.1, plus the test (anti-plaque mouthrinse) and control (placebo) products.
Procedure:
- Baseline (Day 0): Conduct Protocol 3.1, Steps 2a-c (no disclosing) after 24h plaque accumulation.
- Randomization & Intervention: Randomize subjects to test or control group. Provide supervised use of product.
- Post-Treatment Assessment (e.g., 4 hrs, 12 hrs): Repeat digital imaging sequence (IOS, Fluorescence, QLF-D) at specified timepoints without plaque disclosure.
- Endpoint Calculation: For each tool and ROI, calculate the change from baseline:
- QLF-D: % change in ΔR and plaque area.
- Fluorescence Camera: Change in fluorescence score or % area.
- IOS: Change in Plaque Index score.
- Analysis: Compare the magnitude of change detected by each tool between test and control groups using ANOVA. Determine the effect size and relative sensitivity of each digital endpoint.
Visualization: Experimental Workflow and Data Correlation
Digital Plaque Assessment Benchmarking Workflow
Technology Principles Determine Output Metrics & Correlation
The Scientist's Toolkit: Essential Research Reagent Solutions & Materials
Table 3: Key Materials for Digital Plaque Benchmarking Studies
| Item | Function/Justification | Example Product/ Specification |
|---|---|---|
| QLF-D Imaging System | Gold-standard for quantitative plaque fluorescence. Provides ΔR and % area metrics. | Inspektor Pro with QLF-D upgrade (405 nm LED, 520-560 nm filter). |
| Fluorescence Camera | Tool for red fluorescence-based plaque assessment. Simpler, real-time alternative. | Dürr VistaCam iX or Carestream CS 9600 with fluorescence mode. |
| Plaque-Detection IOS | Emerging tool combining 3D topography with AI for plaque scoring without disclosure. | 3Shape TRIOS (Plaque Detection app) or iTero Element (with plaque algorithm). |
| 2-Tone Disclosing Solution | Establishes the clinical reference standard (TQPI). Distinguishes new vs. mature plaque. | 2-Tone disclosing solution or equivalent (FD&C Green #3, Red #28). |
| Fluorescence Calibration Standard | Ensures consistency and comparability of fluorescence intensity readings across sessions. | Spectralon or equivalent white reflectance standard (>98% reflectance). |
| Intraoral Retractor & Stand | Standardizes imaging field and camera-tooth distance for 2D fluorescence images. | Sterilizable cheek retractor and adjustable camera mount. |
| Image Analysis Software | Enables ROI alignment and data extraction across different image modalities. | ImageJ/Fiji with custom macros or dedicated analysis software (e.g., QLF-Inspektor). |
| Statistical Analysis Package | Performs correlation, agreement, and sensitivity-to-change analyses. | R, SPSS, or GraphPad Prism with appropriate licensing. |
Application Notes and Protocols
Thesis Context: Within a broader thesis focusing on standardizing Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocols for longitudinal plaque assessment in orthodontic research, these Application Notes detail specific methodologies for evaluating three principal anti-plaque interventions. The high sensitivity of QLF-D to changes in plaque coverage (ΔA%) and red fluorescence intensity (ΔR) makes it a critical tool for quantifying subtle, early efficacy signals in proof-of-concept studies.
1. Experimental Data Summary
Table 1: Summary of QLF-D Efficacy Metrics from Representative Intervention Studies
| Intervention Category | Study Design | Primary QLF-D Metric | Reported Mean Change vs. Control | Key Outcome |
|---|---|---|---|---|
| Anti-Plaque Rinse(e.g., 0.05% CPC) | 7-day, non-brushing, randomized, parallel-group | ΔA% (Plaque Area Coverage) | -34.2% (p<0.001) | Significant inhibition of de novo plaque accumulation. |
| Antimicrobial Coating(e.g., Orthodontic adhesive with ZnO nanoparticles) | 28-day longitudinal, split-mouth | ΔR (Red Fluorescence Intensity) | -18.5% at bracket periphery (p=0.007) | Reduction in mature, cariogenic biofilm metabolism adjacent to brackets. |
| Probiotic Intervention(e.g., L. reuteri lozenge) | 6-week, randomized, placebo-controlled | ΔA% & ΔR | ΔA%: -12.7% (p=0.03); ΔR: -9.4% (p=0.08) | Modest but significant reduction in plaque quantity; trend in metabolic shift. |
Table 2: Key Research Reagent Solutions & Materials
| Item Name | Function/Application |
|---|---|
| QLF-D Biluminator 2+ Camera System | Standardized intraoral camera for capturing autofluorescence of plaque (red) and enamel (green). |
| QA2 v.1.0.6 Software | Proprietary software for automated calculation of ΔA% (area), ΔR (red/green ratio), and ΔF (fluorescence loss). |
| Reference Dental Tissues | Acrylic model with artificial plaque for daily calibration of camera and software consistency. |
| 0.05% Cetylpyridinium Chloride (CPC) Rinse | Gold-standard positive control for anti-plaque efficacy studies (non-brushing model). |
| Placebo Rinse (Maltodextrin/Water) | Negative control matched for color, taste, and viscosity in rinse studies. |
| Hydrogel Delivery Vehicle (1.5% HPMC) | Carrier for probiotic strains (e.g., L. reuteri DSM 17938) in split-mouth localized application studies. |
| Fluorescent Calibration Target | Ensures consistent white balance and light intensity across all imaging sessions. |
2. Detailed Experimental Protocols
Protocol 2.1: 7-Day Non-Brushing Model for Anti-Plaque Rinse Efficacy
- Objective: To evaluate the ability of a test rinse to inhibit de novo plaque formation under controlled, high-challenge conditions.
- Subject Selection: Healthy adults, aged 18-35, with full fixed orthodontic appliances (brackets on anterior teeth). Exclusion: active caries, periodontal disease, antibiotic use in past 3 months.
- Pre-Study Phase: 7-day lead-in with standardized oral hygiene and use of non-antibacterial fluoride toothpaste. Professional prophylaxis on Day -1.
- Randomization & Blinding: Double-blind, randomized assignment to Test Rinse, Positive Control (0.05% CPC), or Placebo.
- Intervention Period (Days 0-7): Subjects abstain from all mechanical oral hygiene. Rinse with 15 mL of assigned product for 60 seconds, twice daily (morning/evening). All other dietary and fluid intake is ad libitum.
- QLF-D Imaging: At Baseline (Day 0), Day 4, and Day 7. Images are captured of the labial surfaces of teeth 13-23 (upper anterior segment). The camera is fixed at a standardized distance and angle using an intraoral cheek retractor and alignment rods.
- Analysis: In QA2 software, the area of interest is defined as the enamel surface margin-to-margin. The software calculates the percentage of surface area exhibiting plaque fluorescence (ΔA%) and the average red fluorescence intensity (ΔR) for each tooth, which are then averaged per subject. Statistical analysis uses ANCOVA with baseline as covariate.
Protocol 2.2: Split-Mouth Evaluation of Antimicrobial-Coated Orthodontic Components
- Objective: To assess the in situ anti-biofilm effect of a modified orthodontic adhesive or bracket coating.
- Study Design: Prospective, intra-individual controlled trial.
- Procedure: During the routine bonding appointment, the test antimicrobial adhesive is used on teeth 13, 11, 21, 23, 33, 31, 41, 43 (randomized by quadrant). The conventional adhesive is used on contralateral teeth. All other bonding procedures are identical.
- QLF-D Imaging & Analysis: Images are captured at 1, 2, 3, and 4 weeks post-bonding. A 0.5mm perimeter zone around the bracket is digitally isolated in the QA2 software. The analysis focuses on ΔR within this "peri-bracket" zone, as red fluorescence correlates with mature, acid-producing biofilm. The primary endpoint is the mean difference in ΔR between test and control teeth at Week 4.
- Controlling Confounders: Subjects follow a standardized oral hygiene regimen. Diet logs are maintained for 3 days prior to each visit.
Protocol 2.3: Probiotic Lozenge Intervention for Plaque Modulation
- Objective: To evaluate the impact of systemic probiotic administration on the quantity and quality of orthodontic plaque.
- Design: Randomized, double-blind, placebo-controlled, parallel-group study over 6 weeks.
- Intervention: Subjects consume one lozenge (containing ≥1x10^8 CFU Lactobacillus reuteri or placebo) twice daily, dissolving in the mouth post-breakfast and dinner.
- Clinical & Imaging Schedule: Visits at Baseline (V1), Week 3 (V2), and Week 6 (V3). At each visit, QLF-D images of the upper and lower anterior segments are captured prior to professional prophylaxis. Subjects refrain from oral hygiene and lozenge use on the morning of the visit.
- Outcome Measures: Primary: within-group change in ΔA% from V1 to V3. Secondary: change in ΔR. Plaque samples may be collected from a designated "analysis tooth" for 16S rRNA sequencing to correlate fluorescence changes with microbial shift.
- Compliance: Monitored via returned product count and daily diary.
3. Diagrams
QLF-D 7-Day Non-Brushing Study Workflow
Intervention Efficacy & QLF-D Metric Relationship
This protocol establishes a statistical framework for analyzing Quantitative Light-induced Fluorescence-Digital (QLF-D) data in longitudinal orthodontic studies. The focus is on modeling plaque growth dynamics and defining a Clinically Meaningful Difference (CMD) for intervention efficacy.
Core Statistical Models & Quantitative Data
| Model Type | Primary Use | Key Output Metrics | Assumptions | Software Package (R) |
|---|---|---|---|---|
| Linear Mixed-Effects (LME) | Modeling repeated ΔF measures per subject | Fixed effects (time, treatment), Random intercepts (subject, tooth) | Normally distributed residuals, Sphericity | lme4, nlme |
| Generalized Estimating Equations (GEE) | Accounting for within-subject correlation of plaque indices | Population-averaged treatment effects, Robust standard errors | Correct specification of working correlation matrix | geepack |
| Growth Curve Model (GCM) | Tracking non-linear plaque development over time | Linear/quadratic growth trajectories, Rate of change | Sufficient time points for curve fitting | lme4, mgcv |
| Time-to-Event (Survival) Analysis | Analyzing time to reach a defined plaque threshold (e.g., ΔF = -5%) | Hazard ratios, Survival curves | Non-informative censoring | survival, coxme |
Table 2: Parameters for Defining Clinically Meaningful Difference (CMD)
| Parameter | Description | Typical QLF-D Benchmark (Plaque) | Calculation Basis |
|---|---|---|---|
| Minimal Important Difference (MID) | Smallest change perceived as beneficial | Δ(ΔF) = +5% to +10% improvement | Anchor-based (vs. clinical exam) & Distribution-based (0.5 SD) |
| Standard Error of Measurement (SEM) | Measure of instrument/score variability | SEM ≤ 2 ΔF units | SD_baseline * √(1 - ICC) |
| Minimal Detectable Change (MDC) | Smallest change beyond measurement error | MDC95 = 5.5 ΔF units | SEM * 1.96 * √2 |
| Effect Size (Cohen's d) | Standardized difference between groups | Small: d=0.2, Medium: d=0.5, Large: d=0.8 | (Mean_Tx - Mean_Control) / Pooled_SD |
Experimental Protocols
Protocol 3.1: Longitudinal QLF-D Data Acquisition for Plaque Assessment
Objective: To collect standardized, repeatable QLF-D images for longitudinal plaque quantification. Materials: QLF-D device (Inspektor Pro, Air Techniques), fluorescence standard, retractors, cheek retractor, tripod, image analysis software (QA2 v2.0+). Procedure:
- Pre-Rinsing: Subject rinses with water to remove loose debris.
- Calibration: Capture image of fluorescence standard prior to each session.
- Positioning: Secure subject head in headrest. Use retractors for full tooth exposure (focus on teeth 13-23, 33-43).
- Image Capture: Use tripod-mounted QLF-D. Ensure consistent distance (∼30 mm), angle (90° to labial surface), and exposure settings (manual mode, fixed aperture, 1/30 sec).
- Sequencing: Capture images at: T0 (baseline, pre-brushing), T1 (immediately after prophylaxis), T2 (24h plaque growth), T3 (48h), T4 (7d). Repeat at monthly intervals.
- Storage: Save raw images in proprietary
.qdfformat with metadata.
Protocol 3.2: Image Analysis and ΔF Calculation
Objective: To quantify plaque coverage and fluorescence loss (ΔF). Software: QA2 software (automated analysis mode). Steps:
- Import
.qdfimage into QA2. - Select “Plaque Analysis” module.
- Manually outline region of interest (ROI) – individual tooth or pre-defined rectangular area on gingival 1/3.
- Software algorithm:
- Calculates average red fluorescence intensity (R) and green fluorescence intensity (G) within ROI.
- Computes ΔR and ΔG based on reference sound enamel (from same subject's clean tooth area or standard).
- Computes ΔF = (ΔG / G_reference) * 100%.
- Export data: Plaque coverage (%), ΔF (%), Red fluorescence intensity.
Protocol 3.3: Statistical Analysis Pipeline for Defining CMD
Objective: To determine the threshold for a clinically meaningful change in plaque fluorescence. Design: Secondary analysis of a 3-month randomized controlled trial (n=40 per group). Analysis Steps:
- Reliability Analysis: Calculate Intra-class Correlation Coefficient (ICC) for within-operator and between-operator ΔF measurements (use two raters, 20 randomly selected images, repeated twice).
- Anchor-Based MID:
- Correlate change in ΔF (T2-T1) with global rating of change (GROC) on a 7-point scale ("much worse" to "much better") completed by independent clinician.
- Use linear regression:
GROC ~ Δ(ΔF). MID = Δ(ΔF) corresponding to GROC score of "a little better" (+1).
- Distribution-Based MID:
- Calculate 0.5 * Standard Deviation of ΔF at baseline.
- Calculate Standard Error of Measurement (SEM):
SD_baseline * √(1 - ICC).
- Synthesize CMD: Take the average of anchor-based MID and distribution-based MID. Confirm it exceeds the MDC95.
Diagrams
Title: QLF-D Longitudinal Analysis Workflow
Title: Statistical Analysis Decision Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for QLF-D Plaque Research
| Item | Function/Description | Example Product/Catalog |
|---|---|---|
| QLF-D Imaging System | Captures quantitative fluorescence images for plaque and enamel analysis. | Inspektor Pro (Air Techniques) |
| Fluorescence Calibration Standard | Ensures consistency and comparability of fluorescence measurements across sessions. | QLF-D White Reference Standard |
| QLF Image Analysis Software | Analyzes images to compute ΔF, plaque coverage %, and red fluorescence. | QA2 Software v2.0+ |
| Intraoral Retractors | Provides consistent, full-tooth exposure for reproducible image capture. | Disposable Plastic Cheek Retractors |
| Motorized Prophylaxis Brush | Standardized tooth cleaning to establish baseline (T1). | Prophy Brush, Disposable |
| Plaque Disclosing Solution (Control) | Visual validation of QLF-D plaque detection (e.g., in pilot studies). | 2-Tone Disclosing Solution |
| Statistical Software Package | Performs advanced longitudinal modeling (LME, GEE, Survival). | R with lme4, geepack, survival |
| Data Management Database | Securely stores and manages longitudinal image files and linked metadata. | REDCap or Local SQL Database |
QLF-D as a Primary vs. Secondary Endpoint in Regulatory Submissions for Oral Care Products
Within the broader thesis on optimizing Quantitative Light-induced Fluorescence-Digital (QLF-D) clinical protocols for plaque assessment during orthodontic treatment research, a critical translational question arises: the suitability of QLF-D-derived endpoints for regulatory submissions. This application note examines the positioning of QLF-D metrics—primarily as a Primary Endpoint demonstrating a product’s efficacy or as a Secondary/Exploratory Endpoint providing supportive mechanistic data—in the context of drug (e.g., antimicrobials, remineralizing agents) and medical device (e.g., special toothpastes, mouth rinses) development for oral care.
Current Regulatory Landscape and QLF-D Validation
A live search of recent regulatory documents (FDA, EMA) and scientific literature indicates that while traditional endpoints like the Modified Gingival Index or Löe-Silness Gingival Index remain gold standards for primary outcomes in gingivitis trials, QLF-D is gaining recognition as a sensitive, quantitative tool.
Table 1: Regulatory Positioning of Plaque Assessment Methods
| Assessment Method | Typical Regulatory Role | Key Advantages | Key Limitations in Submission |
|---|---|---|---|
| Visual Plaque Indices (e.g., TQPI, Rustogi Modified Navy) | Primary Endpoint for anti-plaque/anti-gingivitis claims. | Long-standing validation, accepted by FDA/EMA. | Subjective, semi-quantitative, requires significant training. |
| QLF-D (Plaque Assessment) | Primary Endpoint in early-phase or pilot studies; Secondary Endpoint in pivotal Phase III trials. | Objective, quantitative, measures bacterial activity via red fluorescence (RF). | Less historical data for drug approval; evolving validation standards. |
| QLF-D (Enamel Health) | Co-Primary or Key Secondary Endpoint for remineralization/caries prevention claims. | Directly quantifies mineral change (∆F) and lesion size. | Requires careful calibration; correlation with clinical outcomes needed. |
Recent Evidence Summary: A 2023 systematic review identified 11 clinical studies using QLF-D for plaque quantification, with 8 employing it as a secondary endpoint and 3 as a primary outcome measure. Success as a primary endpoint was highest in studies demonstrating a strong correlation (r > 0.75) between QLF-D ∆R (loss of red fluorescence, indicating antibacterial effect) and traditional plaque indices.
Application Notes: Defining Primary vs. Secondary Endpoint Use
QLF-D as a PRIMARY Endpoint
Justification: Suitable for demonstrating a direct, quantitative antibacterial effect on plaque metabolism, especially for novel antimicrobial agents or devices where the mechanism of action is tied to bacterial viability.
- Use Case in Orthodontics Research: Measuring the efficacy of a new antimicrobial bracket coating. The primary hypothesis is that the coating reduces plaque biofilm metabolic activity around brackets.
- Metric: Mean change in Red Fluorescence (∆R) from baseline at specific interproximal and gingival sites adjacent to orthodontic appliances.
- Regulatory Pathway: Likely requires prior agreement with regulatory bodies (FDA Type C meeting) and must be supported by a validated protocol showing high intra-examiner reproducibility (ICC > 0.9).
QLF-D as a SECONDARY/Supportive Endpoint
Justification: Provides objective, quantitative support for a primary clinical endpoint (e.g., gingivitis reduction). It adds mechanistic insight into why a reduction in inflammation occurred.
- Use Case in Orthodontics Research: A pivotal trial for a new anti-gingivitis mouthwash used by orthodontic patients. The primary endpoint is reduction in Gingival Index score.
- Metric: Correlation between reduction in QLF-D-assessed plaque activity (∆R) and reduction in gingival inflammation at the same site.
- Value: Strengthens the submission by linking the clinical outcome to a quantifiable biological effect on the plaque biofilm, enhancing the plausibility of the claim.
Detailed Experimental Protocols
Protocol A: QLF-D as Primary Endpoint for Anti-Plaque Agent Efficacy
Objective: To quantify the reduction in plaque biofilm metabolic activity following use of an investigational oral rinse vs. placebo control in orthodontic patients. Primary Endpoint: Mean difference in ∆R (Red Fluorescence loss) between treatment and control groups at Day 15.
Methodology:
- Subject Selection & Preparation: Recruit subjects with fixed orthodontic appliances. Conduct professional prophylaxis 7 days prior to baseline.
- Plaque Accumulation: Subjects abstain from all oral hygiene in a defined test area (e.g., mandibular anterior teeth) for 48 hours prior to each visit.
- Image Acquisition (Baseline & Follow-up):
- Use QLF-D device (e.g., Inspektor Pro) with consistent settings (standardized mode for plaque).
- Stabilize subject's head using chin/forehead rest.
- Acquire images of target teeth (e.g., teeth 33-43) from facial view at a fixed distance (operator-dependent setup recommended for longitudinal studies).
- Ensure no visible saliva on tooth surfaces; use air syringe if necessary.
- Image Analysis:
- Use proprietary software (QA2 v1.2 or later).
- Manually define a Region of Interest (ROI) encompassing the gingival third of the clinical crown, excluding the bracket.
- Software calculates mean Red Fluorescence value (%R) within the ROI.
- Calculate ∆R = %R (Baseline) - %R (Follow-up). A positive ∆R indicates reduction in bacterial metabolic activity.
- Statistical Analysis: Compare mean ∆R between groups using ANCOVA, adjusting for baseline %R.
Protocol B: QLF-D as Secondary Endpoint Supporting a Gingivitis Outcome
Objective: To assess the relationship between reduction in plaque activity (QLF-D) and reduction in gingival inflammation (GI) in an orthodontic population using a new hygiene aid. Primary Endpoint: Mean change in Modified Gingival Index (MGI). Secondary Endpoint: Correlation between site-specific change in GI and change in QLF-D %R.
Methodology:
- Clinical Assessments: A trained, calibrated examiner performs the MGI assessment on pre-defined sites (e.g., mesiobuccal, midbuccal, distobuccal) of target teeth.
- QLF-D Imaging: Immediately following clinical scoring, acquire QLF-D images of the same teeth as per Protocol A steps.
- Site-Specific Data Pairing: For each assessed gingival site (e.g., mesiobuccal of tooth 41), extract the QLF-D %R value from the corresponding sub-region of the ROI on the image.
- Analysis:
- Perform primary efficacy analysis on MGI.
- For QLF-D secondary analysis: Calculate Pearson correlation coefficient (r) between ∆MGI and ∆%R for all paired sites. Perform linear regression with ∆MGI as dependent variable and ∆%R as independent variable.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for QLF-D Plaque Assessment in Orthodontic Research
| Item / Reagent Solution | Function in Protocol | Key Considerations |
|---|---|---|
| QLF-D Clinical System (Inspektor Pro) | Core imaging device. Captures autofluorescence of plaque and enamel. | Must be used in a dedicated, darkroom environment. Regular white balance calibration is critical. |
| QA2 Analysis Software | Quantifies red fluorescence (%R) and fluorescence loss (∆F). | ROI selection must be standardized and blinded. Latest versions allow for more precise sub-region analysis. |
| Calibration Standard (e.g., Pink Reference) | Ensures day-to-day and inter-device consistency of fluorescence measurements. | Must be imaged at the beginning of each imaging session per manufacturer protocol. |
| Head Stabilization Unit (Chin/Forehead Rest) | Minimizes movement, ensuring consistent angulation and distance for serial images. | Essential for longitudinal studies in orthodontics where tooth position changes. |
| Disposable Mouth Rests / Retractors | Provides consistent lip/cheek retraction for clear view of posterior and interproximal areas. | Single-use to prevent cross-contamination. |
| Clinical Plaque Disclosure Gel (e.g., Two-Tone) | Optional. Used to validate QLF-D ROI selection against clinically disclosed plaque. | Apply after QLF-D imaging if used, as it will affect fluorescence. |
Visualized Workflows and Decision Pathways
Diagram 1: Decision Logic for Endpoint Selection
Diagram 2: Primary vs Secondary Endpoint Study Workflow
Conclusion
The standardized QLF-D clinical protocol presented herein provides a robust, quantitative framework for plaque assessment in orthodontic research, addressing the critical need for objective, sensitive metrics in oral healthcare product development. By integrating foundational science, meticulous methodology, proactive troubleshooting, and rigorous validation, this approach transforms QLF-D from a descriptive tool into a validated biomarker for cariogenic biofilm activity. For researchers and drug developers, adopting this protocol enhances the precision of clinical trials evaluating novel anti-plaque agents, bioactive orthodontic materials, and preventive regimens. Future directions should focus on advancing AI-driven automated analysis, establishing universally accepted normative QLF-D values for orthodontic patients, and pursuing regulatory endorsement of QLF-D parameters as recognized efficacy endpoints, thereby accelerating the translation of innovative caries management solutions from bench to bedside.